NICU Genetic Testing Market Size, Trends and Insights By Product & Service (Consumables & Reagents, Instruments, Software/Services), By Test Type (Newborn Screening Panels/Tests, Diagnostic Techniques, Rapid Whole-Genome Sequencing (rWGS), Exome Sequencing, Others, cCGH, FISH, PCR), By Technology (Next-Generation Sequencing (NGS), Tandem Mass Spectrometry), By End-user (Hospitals & NICUs, Specialty Clinics, Diagnostic Laboratories), and By Region - Global Industry Overview, Statistical Data, Competitive Analysis, Share, Outlook, and Forecast 2026 – 2035
Report Snapshot
| Study Period: | 2026-2035 |
| Fastest Growing Market: | Asia Pacific |
| Largest Market: | North America |
Major Players
- Illumina Inc.
- Thermo Fisher Scientific
- Revvity (erstwhile PerkinElmer)
- Agilent Technologies
- Others
Reports Description
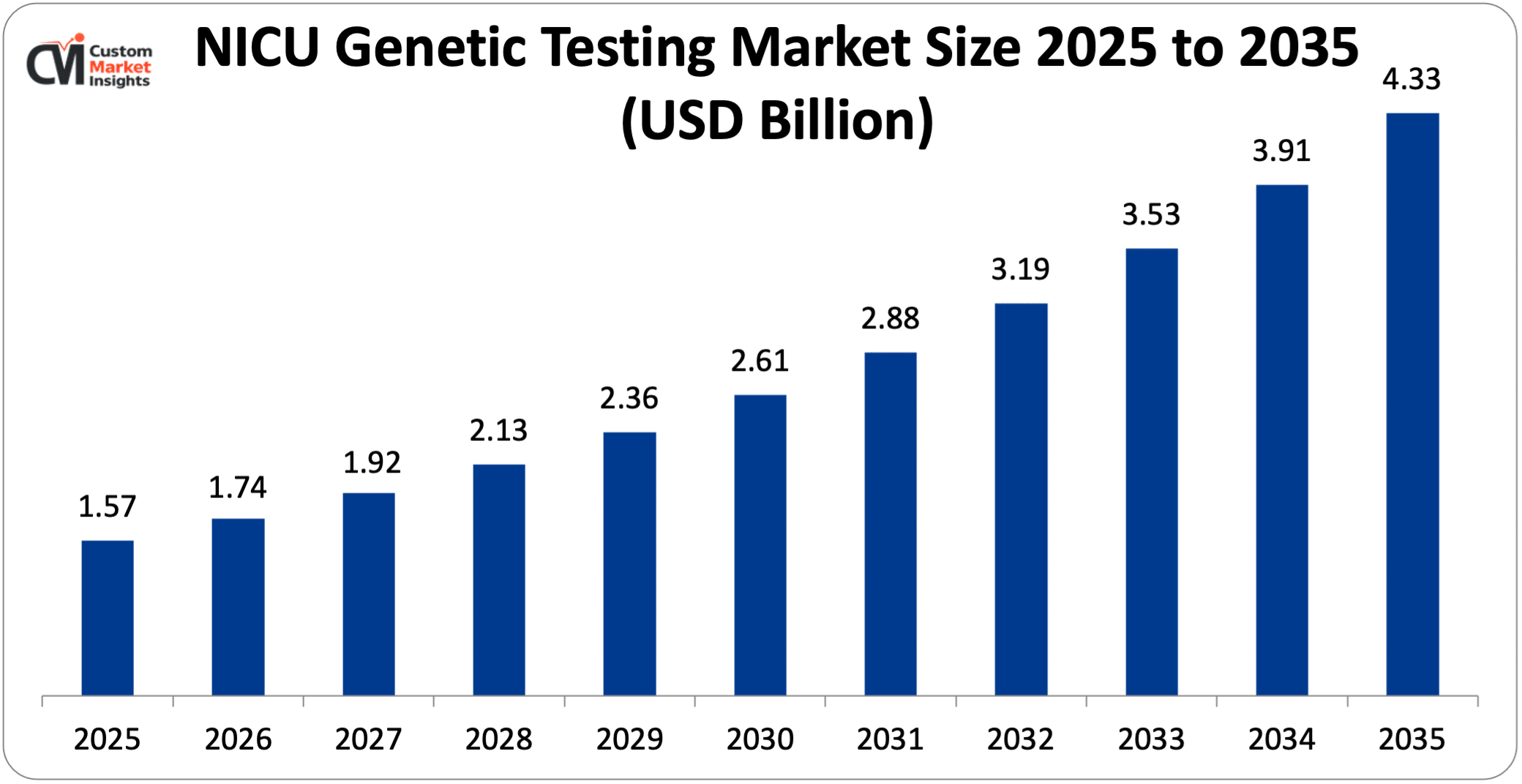
As per the NICU genetic testing market analysis conducted by the CMI team, the NICU genetic testing market is expected to record a CAGR of 10.67% from 2026 to 2035. In 2026, the market size was USD 1.74 Billion. By 2035, the valuation is anticipated to reach USD 4.33 Billion. The market is expanding due to rising demand for advanced sequencing technologies such as rapid whole-genome sequencing (rWGS) across the globe, with North America ruling the roost.
Market Highlights
- North America dominated the NICU genetic testing market in 2025 with 51.73% of the overall share.
- The Asia Pacific is expected to witness the fastest CAGR of 11.5% in the NICU genetic testing market during the forecast period.
- By product & service, consumables & reagents held around 69.37% of the market share by 2025.
- By product & service, the software/services segment is expected to witness the fastest CAGR of 9.34% between 2026 and 2035.
- Through test type, newborn screening panels/tests dominated in 2025 with 58.23% of the overall market share.
- By test type, the diagnostic techniques are expected to witness the fastest CAGR of 10.97% between 2026 and 2035.
- Through technology, next-generation sequencing (NGS) dominated in 2025 with 54.47% of the overall market share.
- By technology, tandem mass spectrometry is expected to witness the fastest CAGR of 9.97% between 2026 and 2035.
- By end-user, hospitals & NICUs dominated in 2025 with 55.64% of the overall market share.
- By end-user, specialty clinics are expected to witness the fastest CAGR of 9.34% between 2026 and 2035.
Significant Growth Factors
- Transition to “Genomics-First” Neonatology
The NICU genetic testing market is set for significant growth in the upcoming period. While conventional genetic testing is already a fast growing market, the traction of genomic services geared toward neonates is paving the way for a new standard of care for infants. Additionally, advancements in genomics are evolving the conventional NICU from “test-based” to “genomics-first” neonatology, thereby enabling a new standard of care in the NICU. The fact that sequencing costs now have decreased considerably has demonstrated that rapid whole-genome sequencing (rWGS) is the first-line diagnostic that offers many economic, logistical and emotional advantages such as the expansion to precision therapeutics such as gene therapies or specialized metabolic diets in the critical first days of life.
- Expansion of Government-mandated NBS (Newborn Screening) Panels
A second big growth area is the expansion of newborn screening tests and better insurance coverage. Laws like the “right to a diagnosis” movements are pushing health systems in the Asia Pacific and Europe to add advanced genetic testing to their screening programs. This is being helped by the use of artificial intelligence to analyze data, which makes it easier for hospitals to handle large amounts of information without needing many more experts. These changes in regulations and technology are turning newborn care units into high-tech centers for testing, which is visibly reducing the number of rare diseases that go undiagnosed.
What are the Major Advancements Changing the NICU Genetic Testing Market Today?
- Transition to Ultra-Rapid Whole Genome Sequencing (urWGS)
The NICU genetic testing market is currently transitioning from traditional genetic testing methods to newer genetic testing technologies, particularly ultra-rapid whole genome sequencing (urWGS). The healthcare industry is witnessing significant advancements, particularly in the field of diagnostics. Rapid diagnostic tests are solutions that save clinicians time and improve the entire healthcare system. Prior to the commencement of clinical treatment, doctors must be informed of an underlying problem in order to provide proper care to patients. The accelerated and effective provision of healthcare during a patient’s first few hours in the NICU is essential toward ensuring certainty for babies who are facing ambiguous omics and genomic diseases.
Newborn sequencing is made possible by ultra-rapid genetic sequencing and the tools it provides, such as open-source tools based on FHIR. Sequencing is made accurate and quick by algorithms supplied by these software solutions. The method involves analyzing each base of the offspring’s genome over the course of about 13.5 to 24 hours in order to produce the findings. For the neonatal department to get results and conduct further genomic analysis, any genome center that engages in sequencing and qualitative research must have a proficient team of bioinformaticians. The newborn sequencing impact is multidimensional. It can fundamentally alter clinical characteristics of care or medical necessity. The tailored treatment that ultrafast sequencing outcomes enable during treatment can significantly expedite therapy, resulting in faster recovery times for patients.
- Impact of AI On NICU Genetic Testing Market
Ai is turning a data collection exercise into a real-time tool to help doctors make decisions. This is happening mainly by solving the problem of interpreting voluminous data. Ai uses machine learning and natural language processing for analyzing a baby’s data and health records. It can now prioritize disease variants in minutes. Experts to review the data manually. This progress determines whole-genome sequencing (rWGS). It allows for results within 24 hours. Doctors can start life-saving treatments or surgeries before permanent damage occurs. Ai is also used for “phenotyping” and predictive modeling. These tools help identify babies who need testing at earlier stages. This reduces hospital stays.
Category Wise insights
By Product & Service
- Why are consumables & reagents dominating the NICU genetic testing market?
The NICU genetic testing market landscape depicts the dominance of the consumables and reagents segment owing to its recurring costs. This segment is likely to account for nearly two-thirds of the total market share; pair it with the wide range of applications of consumables such as routine metabolic screens and complex genomic sequences. For decades, companies like Illumina and Thermo Fisher have been putting in efforts to establish the closed-system model. As a result, these companies have developed an ecosystem around their respective proprietary consumables and have been reaping substantial profits ever since. With the growing focus on driving consumable adoption, the NICU genetic testing market is likely to thrive in the years to come. Several factors contribute to the dominance of consumables and reagents, such as government mandates, global testing volume surges, and high-throughput next-generation sequencing.
By Test Type
- How are newborn screening panels/tests leading the NICU genetic testing market?
Newborn screening panels are among the most important public health mandates made universal. Nearly every infant born undergoes standardized testing of samples of blood and urine, and in some cases this blood testing will extend to screening for genetic disease. As a result, this has created a platform to create stable revenue for the supply of consumables and reagents that are used for testing. These newborn screening panels have reported to be foundational for the early detection of some conditions such as cystic fibrosis and sickle cell disease.
In more symptomatic cases, these panels have allowed for advanced sequencing to accurately detect complex mutations. Newborn screening panels dominate in both – sample volume and market value in terms of the various segments on the market. In terms of volume, samples of blood are used for newborn screening and diagnostic sequencing. Urine and tissue samples can be used to diagnose inborn errors of metabolism (IEMs). In terms of market value, next-generation sequencing will continue to be incorporated into these newborn screening programs.
By Technology
- Why does Next-Generation Sequencing (NGS) segment dominate the NICU genetic testing market?
DNA, which contains complete information about all living organisms, comprises a long chain of chemical shapes (including a, g, c, t) coded as a language. The order of the letters, or sequence of these chemical bases, determines what is built from the DNA. Next-generation sequencing (NGS) of DNA makes such sequencing a lot faster and cheaper, enabling comprehensive genome/exome (DNA across the entire genome or only the parts of important coding genes) testing that reveals the genetic information (or coding) of an individual. This genetic information could be applied in various contexts. When used to replace traditional single-gene testing of DNA, the application can be seen in wide-ranging areas such as agriculture, clinical research, clinical laboratories, or direct-to-consumer. A relatively new area of application is rapid whole-genome sequencing (rWGS) for identifying rare genetic conditions in critically unwell babies in NICUs. The technology can identify rare genetic conditions responsible for a baby’s illness in under 48 hours, providing definitive answers for around 40% of critically ill infants presenting in an acute clinical setting.
By End-user
- Why are hospitals & NICUs leading the NICU genetic testing market?
Hospitals and NICUs are often the primary care hub for many neonates during their first days to weeks of life. Equipped with specialized medical infrastructure and the medical personnel required to care for these high-acuity patients, it stands to reason that neonatal-genomic initiatives are most successfully undertaken in these environments. Implementation of these initiatives will usually necessitate a restructured workflow and creation of multidisciplinary teams that include neonatologists, genetic counselors, clinical microbiologists, and any other providers from additional necessary specialties, including infectious disease experts, metabolic disease experts, pharmacists, nutritionists, etc. In this structured environment, rWGS can be rapidly integrated into the emergency framework of genetic testing that is already part of the specified NICU protocols. Adequate establishment of these protocols is vital; not only does rWGS have a notable team of medical experts working within and among the different departments, but it also facilitates an expedited start to the testing and informs parents of the condition of their child.
Report Scope
| Feature of the Report | Details |
| Market Size in 2026 | USD 1.74 Billion |
| Projected Market Size in 2035 | USD 4.33 Billion |
| Market Size in 2025 | USD 1.57 Billion |
| CAGR Growth Rate | 10.67% CAGR |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Key Segment | By Product & Service, Test Type, Technology, End-user and Region |
| Report Coverage | Revenue Estimation and Forecast, Company Profile, Competitive Landscape, Growth Factors and Recent Trends |
| Regional Scope | North America, Europe, Asia Pacific, Middle East & Africa, and South & Central America |
| Buying Options | Request tailored purchasing options to fulfil your requirements for research. |
Regional Analysis
How big is North America’s NICU genetic testing market size?
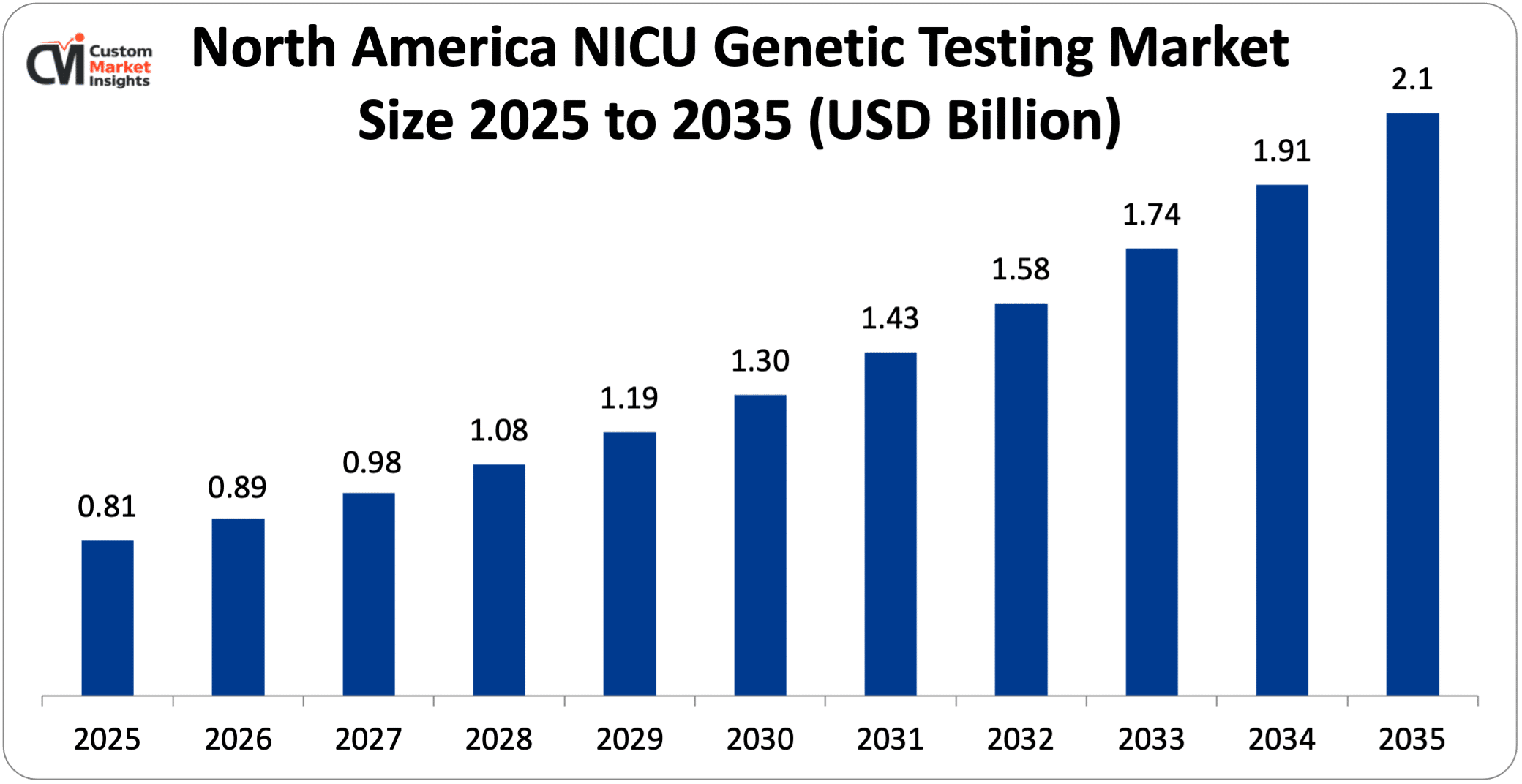
North America’s NICU genetic testing market was worth USD 0.81 Billion in 2025 and is expected to reach USD 2.1 Billion by 2035 at a CAGR of 9.99% between 2025 and 2035.
Why did North America Dominate the NICU Genetic Testing Market in 2025?
North America led the NICU genetic testing market due to the presence of advanced healthcare infrastructure and early integration of rapid whole-genome sequencing (rWGS) in neonatology. Also, reimbursement programs for WGS in NICU cases such as California’s Medi-Cal that provide coverage for high-complexity genomic testing are contributing to North America’s dominance in the global NICU genetic testing market. The presence of the major players providing the next-generation sequencing platforms, such as Illumina, Thermo Fisher, and GeneDx, along with access to AI-driven interpretation tools, is driving the growth in North America.
What is the Size of the U.S. NICU Genetic Testing Market?
The market size of U.S. NICU genetic testing was USD 0.62 Billion in 2025 and is expected to reach USD 1.58 Billion in 2035, witnessing a CAGR of 9.83% between 2026 and 2035.
U.S. NICU Genetic Testing Market Trends
The U.S. neonatal intensive care unit (NICU) genetic testing market is expected to witness noticeable growth by 2035. The implementation of rapid whole-genome sequencing (rWGS) for sick infants is becoming a key trend in the NICU genetic testing market. The California-based healthcare center’s implementation of the 21st century cures act is anticipated to drive NICU testing. Moreover, similar healthcare centers across the globe, like California’s project baby bear are also anticipated to drive the expansion of genetic testing in the NICU market. However, the lack of skilled professionals who can study exomes and genomes is anticipated to hinder the expansion of the NICU genetic testing market. In addition, the introduction of AI-driven interpretation platforms to shorten time-to-diagnosis to under 24 hours is anticipated to boost the U.S. NICU genetic testing market. The integration of pharmacogenomics into NICU services helps to identify the right drug and dose for the newborn patient, which is anticipated to drive the NICU genetic testing market. The emergence of various players for newborn genetic testing is contributing toward the rise in the revenue from the NICU genetic testing market.
Why is the Asia Pacific experiencing the fastest growth in the NICU genetic testing market?
The NICU genetic testing market in the Asia Pacific includes china, india, and economies where newborn screening processes are enlarging. Investment into high-throughput structure and adoption of next-generation sequencing are bound to generate a sizable revenue. Growing middle-class population with improving pediatric medical treatment and increasing parental knowledge of the merits and causes for precision medication will be the driving aspects included in the increase in certain parts of the NICU genetic checking market. Economies like india and china are making strides in the form of presenting advancements in the NICU genetic screening market.
China NICU Genetic Testing Market Trends
Driven by government initiatives, technological advancements, and the pursuit of a better quality of life, china’s NICU genetic testing market continues to grow rapidly. In 2016, china’s government issued the “healthy china 2030” mandate, which required the ministry of health to set up a nationwide network for genetic testing. For instance, the national neonatal screening network is being expanded to include NICU genetic testing based on next-generation sequencing (NGS) technologies. In addition, provinces that have high incidences of congenital disorders or experience difficulty with providing them with reasonable medical attention will receive provincial subsidies for rapid whole-genome sequencing (rWGS) on infants.
As the economy shifts to NGS-based testing, hospitals can simultaneously test infant patients for several genetic mutations at once. With the average cost of rWGS for 1-2 patients equal to that of traditional sanger sequencing (TSS) for 50 infants, provincial healthcare schemes are expanding in order to ensure that the entire population benefits from the country’s technological advancements. New initiatives are leveraging the country’s world-class machine learning algorithms like AI-powered automated variant interpretation, as well as 5G-enabled “tele-neonatology,” to close the diagnostic gap between urban and rural hospitals.
Where does Europe stand with respect to the NICU genetic testing market?
The European market for NICU genetic testing is being propelled by the increasing knowledge of the applications of genomic-first diagnostics. One such major factor that is pushing the growth of the industry in other parts of the region is the successful formation of the UK NHS genomic medicine service that is revolutionizing the neonatal care in the country in an unprecedented manner. These initiatives are being assisted by several governmental initiatives in the area of genomic testing. As next-generation sequencing is being incorporated into more advanced sequencing technologies, it is expected that initiatives like NHS genomic medicine services are expected to present the next future step, as “whole-genome sequencing” will be an essential component in the healthcare of infants and parents. The establishment of harmonized policies and regulations for encouraging the implementation of next-generation sequencing reimbursement and the implementation of genomic initiatives in multiple regions are some of the major factors that are contributing to the growth of the business.
Germany NICU Genetic Testing Market Trends
The significance of the market for genetic testing in newborns is steadily increasing in Germany – both – in terms of economic strength and in terms of the healthcare system. The good use of modern and developed technical possibilities has offered the field of genetics the opportunity to quickly examine and identify individual gene mutations in newborns in order to carry out early therapy. As such, several vendors are providing the rapid whole-genome sequencing (rWGS), which could provide a diagnostic answer to a complex clinical picture within a manageable time frame. The results of these steps are reflected in improved patient care. On the one hand, the more structures and processes are adapted to the new emphasis on precision medicine – as is now being urged in connection with the massive promotion of nuclear medicine and genetic testing in adults – the more effectively these methods will dovetail with established and mature high-throughput methods in order to work consistently and sustainably.
Where is the Middle East & Africa regarding NICU genetic testing market?
In the middle east region, especially among wealthy gulf countries, many patients suffer from rare genetic disorders arising from the practice of having large families and consanguinity. Therefore, regional governments are investing handsomely in establishing genomic databases that combine genetic information of the population, family trees, and geographical location. Countries in the region are also leading in performing and launching global centers of excellence in fields like rapid whole-genome sequencing that are crucial for understanding rare genetic disorders. For example, starting from July 2014, Saudi Arabia and the UAE are turning out to be global leaders in rapid whole-genome sequencing. Various hospitals and health organizations in these economies are investing in developing state-of-the-art genomic facilities to analyze patients’ conditions in a cost-efficient manner and better aid in recognizing the occurrence of rare genetic disorders. The testing results can help provide precision treatments that are otherwise complex and time-consuming. Also, advanced capabilities in genetic testing can help educate the population about the prevalence of genetic disorders in families and communities. Furthermore, in April 2018, the Abu Dhabi health services co. Announced a partnership with Illumina for the establishment of a genomic data center in Abu Dhabi.
However, the African nations are focusing on establishing cost-effective newborn screening for genetic disorders like sickle cell disease. Several governments and health ministries are also launching initiatives and promoting partnerships with research institutions to strengthen awareness among new parents. Even though they face major infrastructural gaps, the sub-Saharan African nations have been recognized as having huge growth potential owing to the falling costs of next-generation sequencing.
Brazil NICU Genetic Testing Market Trends
While the national neonatal screening program (PNTN) creates a robust and long-standing newborn screening framework in brazil, it is experiencing a significant leap since the last few years, particularly with the gradual transition to next-generation sequencing (NGS) panels. Brazil’s newborn screening is still significantly behind its mature market counterparts, where new high-precision methodologies are incorporated almost in real-time. However, investment in the strategy of expanding the portfolio of genetic tests included in the public health system has been sporadic and focused on specific tests for rare diseases that have existing and proven methodologies at lower costs, resulting in the persistence of major gaps in the prenatal and neonatal periods.
This model is currently applied in a race against time for the incorporation of genetic tests in the public healthcare system. Several groups from state health secretariats across brazil are reviewing methodologies for variant interpretation to accommodate precision neonatology and are promoting the incorporation of telegenetics for this purpose.
Top players in the NICU Genetic Testing Market and their Offerings
- Illumina Inc.
- Thermo Fisher Scientific
- Revvity (erstwhile PerkinElmer)
- Agilent Technologies
- Others
Key Developments
The NICU genetic testing market has experienced considerable changes in the last few years as the market players are trying to diversify their technological aspects and develop product portfolios using strategic approaches.
- In November 2024, ProPhase Labs, Inc. announced that they had introduced DNA Complete, Inc., a 100% owned subsidiary that offers D2C DNA tests.
These strategic measures have enabled the companies to reinforce their competitive positions, increase the product line, boost their technological competencies, and also seize growth opportunities in the fast-growing NICU genetic testing market.
The NICU Genetic Testing Market is segmented as follows:
By Product & Service
- Consumables & Reagents
- Instruments
- Software/Services
By Test Type
- Newborn Screening Panels/Tests
- Diagnostic Techniques
- Rapid Whole-Genome Sequencing (rWGS)
- Exome Sequencing
- Others
- cCGH
- FISH
- PCR
By Technology
- Next-Generation Sequencing (NGS)
- Tandem Mass Spectrometry
By End-user
- Hospitals & NICUs
- Specialty Clinics
- Diagnostic Laboratories
Regional Coverage:
North America
- U.S.
- Canada
- Mexico
- Rest of North America
Europe
- Germany
- France
- U.K.
- Russia
- Italy
- Spain
- Netherlands
- Rest of Europe
Asia Pacific
- China
- Japan
- India
- New Zealand
- Australia
- South Korea
- Taiwan
- Rest of Asia Pacific
The Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of the Middle East & Africa
Latin America
- Brazil
- Argentina
- Rest of Latin America
Table of Contents
- Chapter 1. Report Introduction
- 1.1. Report Description
- 1.1.1. Purpose of the Report
- 1.1.2. USP & Key Offerings
- 1.2. Key Benefits For Stakeholders
- 1.3. Target Audience
- 1.4. Report Scope
- 1.1. Report Description
- Chapter 2. Market Overview
- 2.1. Report Scope (Segments And Key Players)
- 2.1.1. NICU Genetic Testing by Segments
- 2.1.2. NICU Genetic Testing by Region
- 2.2. Executive Summary
- 2.2.1. Market Size & Forecast
- 2.2.2. NICU Genetic Testing Market Attractiveness Analysis, By Product & Service
- 2.2.3. NICU Genetic Testing Market Attractiveness Analysis, By Test Type
- 2.2.4. NICU Genetic Testing Market Attractiveness Analysis, By Technology
- 2.2.5. NICU Genetic Testing Market Attractiveness Analysis, By End-user
- 2.1. Report Scope (Segments And Key Players)
- Chapter 3. Market Dynamics (DRO)
- 3.1. Market Drivers
- 3.1.1. Transition to “Genomics-First” Neonatology
- 3.1.2. Expansion of Government-mandated NBS (Newborn Screening) Panels
- 3.2. Market Restraints
- 3.3. Market Opportunities
- 3.5. Pestle Analysis
- 3.6. Porter Forces Analysis
- 3.7. Technology Roadmap
- 3.8. Value Chain Analysis
- 3.9. Government Policy Impact Analysis
- 3.10. Pricing Analysis
- 3.1. Market Drivers
- Chapter 4. NICU Genetic Testing Market – By Product & Service
- 4.1. Product & Service Market Overview, By Product & Service Segment
- 4.1.1. NICU Genetic Testing Market Revenue Share, By Product & Service , 2025 & 2035
- 4.1.2. Consumables & Reagents
- 4.1.3. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 4.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.5. Key Market Trends, Growth Factors, & Opportunities
- 4.1.6. Instruments
- 4.1.7. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 4.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.9. Key Market Trends, Growth Factors, & Opportunities
- 4.1.10. Software/Services
- 4.1.11. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 4.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.13. Key Market Trends, Growth Factors, & Opportunities
- 4.1. Product & Service Market Overview, By Product & Service Segment
- Chapter 5. NICU Genetic Testing Market – By Test Type
- 5.1. Test Type Market Overview, By Test Type Segment
- 5.1.1. NICU Genetic Testing Market Revenue Share, By Test Type, 2025 & 2035
- 5.1.2. Newborn Screening Panels/Tests
- 5.1.3. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 5.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.5. Key Market Trends, Growth Factors, & Opportunities
- 5.1.6. Diagnostic Techniques
- 5.1.6.1. Rapid Whole-Genome Sequencing (rWGS)
- 5.1.6.2. Exome Sequencing
- 5.1.7. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 5.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.9. Key Market Trends, Growth Factors, & Opportunities
- 5.1.10. Others
- 5.1.10.1. cCGH
- 5.1.10.2. FISH
- 5.1.10.3. PCR
- 5.1.11. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 5.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.13. Key Market Trends, Growth Factors, & Opportunities
- 5.1. Test Type Market Overview, By Test Type Segment
- Chapter 6. NICU Genetic Testing Market – By Technology
- 6.1. Technology Market Overview, By Technology Segment
- 6.1.1. NICU Genetic Testing Market Revenue Share, By Technology , 2025 & 2035
- 6.1.2. Next-Generation Sequencing (NGS)
- 6.1.3. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 6.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.5. Key Market Trends, Growth Factors, & Opportunities
- 6.1.6. Tandem Mass Spectrometry
- 6.1.7. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 6.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.9. Key Market Trends, Growth Factors, & Opportunities
- 6.1. Technology Market Overview, By Technology Segment
- Chapter 7. NICU Genetic Testing Market – By End-user
- 7.1. End-user Market Overview, By End-user Segment
- 7.1.1. NICU Genetic Testing Market Revenue Share, By End-user , 2025 & 2035
- 7.1.2. Hospitals & NICUs
- 7.1.3. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 7.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 7.1.5. Key Market Trends, Growth Factors, & Opportunities
- 7.1.6. Specialty Clinics
- 7.1.7. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 7.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 7.1.9. Key Market Trends, Growth Factors, & Opportunities
- 7.1.10. Diagnostic Laboratories
- 7.1.11. NICU Genetic Testing Share Forecast, By Region (USD Billion)
- 7.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 7.1.13. Key Market Trends, Growth Factors, & Opportunities
- 7.1. End-user Market Overview, By End-user Segment
- Chapter 8. NICU Genetic Testing Market – Regional Analysis
- 8.1. NICU Genetic Testing Market Overview, By Region Segment
- 8.1.1. Global NICU Genetic Testing Market Revenue Share, By Region, 2025 & 2035
- 8.1.2. Global NICU Genetic Testing Market Revenue, By Region, 2026 – 2035 (USD Billion)
- 8.1.3. Global NICU Genetic Testing Market Revenue, By Product & Service , 2026 – 2035
- 8.1.4. Global NICU Genetic Testing Market Revenue, By Test Type, 2026 – 2035
- 8.1.5. Global NICU Genetic Testing Market Revenue, By Technology , 2026 – 2035
- 8.1.6. Global NICU Genetic Testing Market Revenue, By End-user , 2026 – 2035
- 8.2. North America
- 8.2.1. North America NICU Genetic Testing Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 8.2.2. North America NICU Genetic Testing Market Revenue, By Product & Service , 2026 – 2035
- 8.2.3. North America NICU Genetic Testing Market Revenue, By Test Type, 2026 – 2035
- 8.2.4. North America NICU Genetic Testing Market Revenue, By Technology , 2026 – 2035
- 8.2.5. North America NICU Genetic Testing Market Revenue, By End-user , 2026 – 2035
- 8.2.6. U.S. NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.2.7. Canada NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.2.8. Mexico NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.2.9. Rest of North America NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.3. Europe
- 8.3.1. Europe NICU Genetic Testing Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 8.3.2. Europe NICU Genetic Testing Market Revenue, By Product & Service , 2026 – 2035
- 8.3.3. Europe NICU Genetic Testing Market Revenue, By Test Type, 2026 – 2035
- 8.3.4. Europe NICU Genetic Testing Market Revenue, By Technology , 2026 – 2035
- 8.3.5. Europe NICU Genetic Testing Market Revenue, By End-user , 2026 – 2035
- 8.3.6. Germany NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.7. France NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.8. U.K. NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.9. Russia NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.10. Italy NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.11. Spain NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.12. Netherlands NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.13. Rest of Europe NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.4. Asia Pacific
- 8.4.1. Asia Pacific NICU Genetic Testing Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 8.4.2. Asia Pacific NICU Genetic Testing Market Revenue, By Product & Service , 2026 – 2035
- 8.4.3. Asia Pacific NICU Genetic Testing Market Revenue, By Test Type, 2026 – 2035
- 8.4.4. Asia Pacific NICU Genetic Testing Market Revenue, By Technology , 2026 – 2035
- 8.4.5. Asia Pacific NICU Genetic Testing Market Revenue, By End-user , 2026 – 2035
- 8.4.6. China NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.7. Japan NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.8. India NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.9. New Zealand NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.10. Australia NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.11. South Korea NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.12. Taiwan NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.13. Rest of Asia Pacific NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.5. The Middle-East and Africa
- 8.5.1. The Middle-East and Africa NICU Genetic Testing Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 8.5.2. The Middle-East and Africa NICU Genetic Testing Market Revenue, By Product & Service , 2026 – 2035
- 8.5.3. The Middle-East and Africa NICU Genetic Testing Market Revenue, By Test Type, 2026 – 2035
- 8.5.4. The Middle-East and Africa NICU Genetic Testing Market Revenue, By Technology , 2026 – 2035
- 8.5.5. The Middle-East and Africa NICU Genetic Testing Market Revenue, By End-user , 2026 – 2035
- 8.5.6. Saudi Arabia NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.5.7. UAE NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.5.8. Egypt NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.5.9. Kuwait NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.5.10. South Africa NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.5.11. Rest of the Middle East & Africa NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.6. Latin America
- 8.6.1. Latin America NICU Genetic Testing Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 8.6.2. Latin America NICU Genetic Testing Market Revenue, By Product & Service , 2026 – 2035
- 8.6.3. Latin America NICU Genetic Testing Market Revenue, By Test Type, 2026 – 2035
- 8.6.4. Latin America NICU Genetic Testing Market Revenue, By Technology , 2026 – 2035
- 8.6.5. Latin America NICU Genetic Testing Market Revenue, By End-user , 2026 – 2035
- 8.6.6. Brazil NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.6.7. Argentina NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.6.8. Rest of Latin America NICU Genetic Testing Market Revenue, 2026 – 2035 (USD Billion)
- 8.1. NICU Genetic Testing Market Overview, By Region Segment
- Chapter 9. Competitive Landscape
- 9.1. Company Market Share Analysis – 2025
- 9.1.1. Global NICU Genetic Testing Market: Company Market Share, 2025
- 9.2. Global NICU Genetic Testing Market Company Market Share, 2024
- 9.1. Company Market Share Analysis – 2025
- Chapter 10. Company Profiles
- 10.1. Illumina Inc.
- 10.1.1. Company Overview
- 10.1.2. Key Executives
- 10.1.3. Product Portfolio
- 10.1.4. Financial Overview
- 10.1.5. Operating Business Segments
- 10.1.6. Business Performance
- 10.1.7. Recent Developments
- 10.2. Thermo Fisher Scientific
- 10.3. Revvity (erstwhile PerkinElmer)
- 10.4. Agilent Technologies
- 10.5. Others.
- 10.1. Illumina Inc.
- Chapter 11. Research Methodology
- 11.1. Research Methodology
- 11.2. Secondary Research
- 11.3. Primary Research
- 11.3.1. Analyst Tools and Models
- 11.4. Research Limitations
- 11.5. Assumptions
- 11.6. Insights From Primary Respondents
- 11.7. Why Healthcare Foresights
- Chapter 12. Standard Report Commercials & Add-Ons
- 12.1. Customization Options
- 12.2. Subscription Module For Market Research Reports
- 12.3. Client Testimonials
- Chapter 13. List Of Figures
- 13.1. Figures No 1 to 34
- Chapter 14. List Of Tables
- 14.1. Tables No 1 to 51
Prominent Player
- Illumina Inc.
- Thermo Fisher Scientific
- Revvity (erstwhile PerkinElmer)
- Agilent Technologies
- Others
FAQs
The key players in the market are Illumina Inc., Thermo Fisher Scientific, Revvity (erstwhile PerkinElmer), Agilent Technologies, Others.
The government regulations regarding NICU genetic testing are bound to assist the growth of the market due to the high public health standards set. As mandated by the recommended uniform screening panel (RUSP) report by the department of health and human services (HHS), various genetic and metabolic disorders are subject to mandatory testing. Genetic tests are offered to families who are reluctant to undergo them.
The genetic information nondiscrimination act (GINA), which forbids employers and health insurers from treating patients differently based on their genetic information, and CLIA laboratory certifications, which guarantee an ethically sound framework for the tests and also assure that the results have high analytical validity, are additional regulations and compliances the NICU genetic testing market must follow. Tests for the diagnosis of specific ailments or treatment options are administered to newborns in the NICU.
Severe types of genetic and hereditary disorders that result in severe disabilities are frequently picked up by the NICU tests that newborns undergo. The market for NICU genetic testing is predicted to rise significantly in the next few years, particularly in emerging economies. For instance, there is a valuable opportunity for the NICU genetic testing market in india due to the passing of the national policy for rare diseases, which promotes universal screening for various metabolic objectives and disorders.
Although targeted sequencing and rWGS now deliver greater clinical value, the high price point of testing remains a barrier to adoption for many healthcare systems across the globe. However, as the growing evidence base demonstrating the clinical and economic value of genetic testing continues to mount, demand among healthcare systems will grow to such a point that they will be unable to ignore the value of implementing testing as a routine part of targeted therapies. For example, while an individual rWGS test may cost $5,000, when used to rule out the possibility of undiagnosed genetic disease causing infant morbidity, such early identification can prevent $50,000 to $100,000 in total facility NICU care costs. Accordingly, a shift in perception of the cost of genetic sequencing from an upfront test cost to ongoing treatment savings is needed for NICU genetic testing markets to rapidly grow globally.
In response to increasing demand amongst healthcare systems and the growing prevalence of rWGS technology, the range of tiered pricing models is rapidly increasing. The scalable nature of next-generation sequencing technologies makes them suitable to this type of tiered pricing model, as the expenses of running a genetic test will decrease as the technology rapidly becomes self-running and less labor-intensive.
The market for NICU genetic testing is expected to witness a significant growth of about USD 4.33 Billion in the year 2035 with a CAGR of 10.67% between the years 2026 and 2035 due to growing incidences of rare genetic disorders followed by faster adoption of NGS (next-generation sequencing) that allows for various life-saving diagnoses in the critically ill infants within 24-48 hours.
North America is projected to dominate the NICU genetic testing market during the forecast period owing to factors such as advanced healthcare infrastructure, faster implementation of rWGS (rapid whole-genome sequencing) as the new standard of care, and robust reimbursement frameworks. For instance, Medicare and private insurance codes covering rWGS allow reimbursement for patients who meet medical necessity criteria. The exceptional reimbursement framework for genetic testing in North America is a vital factor contributing to the region’s growth. Industry leaders such as Illumina, Thermo Fisher, and GeneDx have contributed to the development of advanced genetic tests in North America. Additionally, the high awareness of physicians related to the benefits of using precision medicine in the treatment of congenital disorders should further promote keen industry growth.
Neonatal genetic testing is the fastest-growing segment of the commercial footprint in the Asia Pacific. Asia is home to the biggest share of the global population, with a disproportionately massive birth pool. The combination of these two factors implies that there is a compelling opportunity for the growth of neonatal genetics. The substantial rise in the number of patients requiring genetic testing can be addressed in a timely manner only if there is an updated government healthcare policy and proper government healthcare facilities. In the Asia Pacific, modernizing government healthcare is a priority for economies looking to develop a well-balanced society with adequate government healthcare.
One of the key focuses of modernizing healthcare systems is to reduce infant mortality, and newborn screening in the Asia-Pacific has been identified as the most feasible solution to achieve this goal. Creating a platform in this neonatal genetics market has already been set up by Chinese and Indian economies. These economies are significantly expanding their newborn screening capabilities and establishing high-end high-throughput infrastructure in metropolitan areas to provide efficient testing. The respective governments in these two countries are ensuring doctors have easy accessibility to the tests, and the government is also investing to improve diagnostic access, creating a “leapfrog” effect to bridge the accessibility gap between rural areas and major metropolitan surgical units.
The neonatal intensive care unit (NICU) genetic testing market is expected to see rapid growth over the forecast period due to the increasing demand for neonatal diagnostics owing to rising incidence of genetic disorders and increasing maternal age. According to the American College of Obstetricians and Gynecologists (ACOG), maternal age is defined as a woman being 35 years old and above at the time of delivery, and those women of age 35 and older have a greater chance of delivery of babies with chromosomal abnormalities, genetic lesions, and rare congenital malformations. The NICU is a special department in the hospital or medical centers that takes care of the most vulnerable patients, such as preterm and critically ill newborns.
The introduction of technological advancements and the development of new tests have changed the approach to the diagnosis of genetic disorders in the NICU. Next-generation sequencing (NGS) is a growing tool in NICU genetic testing. Rapid whole-genome sequencing (RWGS) is a recent technological advancement in genetic analysis that helps healthcare professionals in diagnosing genetic disorders and the detection of possible outcomes of the disorder in newborns. Such advancements in genetic testing have led to a reduction of the time to find a diagnosis by utilizing NGS and rWGS of the neonatal intensive care unit (NICU), thus reducing the diagnostic odyssey and promoting market growth.
Furthermore, increasing favorable government mandates supporting expanded newborn screening are also expected to foster the NICU genetic testing market growth. For instance, to promote newborn screening, the U.S. Food and Drug Administration (FDA) launched the newborn screening (NBS) program aimed at identifying infants with serious health conditions at birth so that treatment can start early.
Newborn screening is done within the first few days and usually involves a tiny blood sample from the baby’s heel. Screening is done for conditions that can cause serious problems like brain damage, illness, and death if treatment is not started right away. This NBS program helps promote accurate diagnosis and early treatment, and the newborn screening program in developed countries is expanded to support the availability of new diagnostic methodologies.