Bile Acid Sequestrants Market Size, Trends and Insights By Drug Type (Cholestyramine (Prevalite, Questran, Questran Lite), Colesevelam (Welchol), Colestipol (Colestid, Flavored Colestid), Others), By Indication (Hypercholesterolemia, Primary Hypercholesterolemia, Familial Hypercholesterolemia (Heterozygous), Mixed Dyslipidemia, Type 2 Diabetes Mellitus (Glycemic Control Adjunct), Cholestasis and Biliary Obstruction (Pruritus Relief), Bile Acid Malabsorption and Diarrhea, Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Drug Stores, Others), and By Region - Global Industry Overview, Statistical Data, Competitive Analysis, Share, Outlook, and Forecast 2026 – 2035
Report Snapshot
| Study Period: | |
| Fastest Growing Market: | |
| Largest Market: |
Major Players
Reports Description
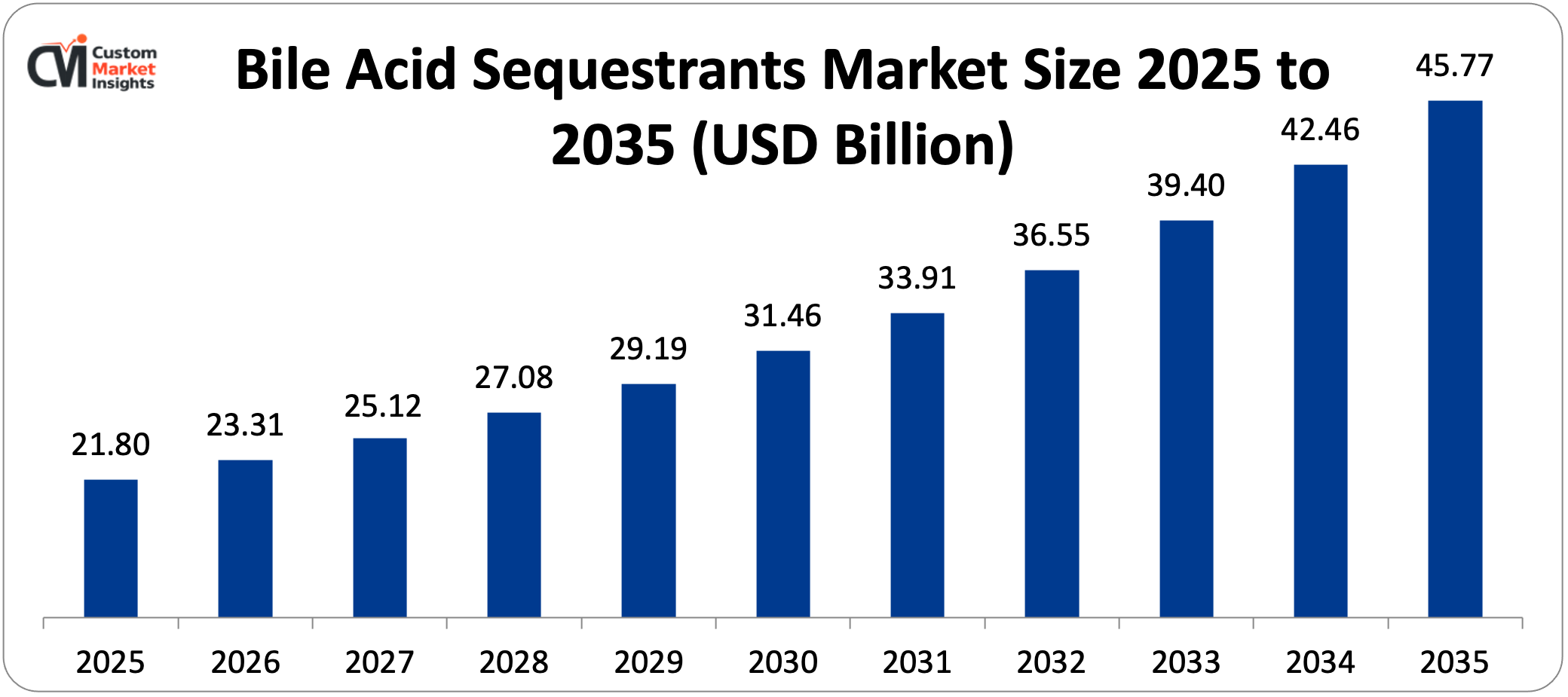
The market size of global bile acid sequestrants has surpassed USD 21.8 billion in 2025 and is expected to capture a CAGR of more than 7.7% to hit above USD 45.77 billion in revenue in 2035. It is projected that the market in 2026 will be USD 23.31 billion. The increasing prevalence of hypercholesterolemia and cardiovascular diseases, ageing population, which is prone to lipid disorders, increased use of combination lipid-lowering therapies, greater clinical indications of bile acid sequestrants beyond cholesterol management, and increased awareness of cardiovascular risk reduction are all driving market expansion.
Market Highlight
- North America is expected to capture the largest portion of the bile acid sequestrants market by 2035 due to the popularity of cholestyramine in treating the symptoms of pruritus in patients with liver cirrhosis.
- Europe is expected to grow significantly by 2026-2035, based on the increase in diabetes rates and greater demand for sophisticated hyperlipidemia treatment.
- By 2035, the colestipol segment will have a market share of 35% of bile acid sequestrants due to its wide application in lowering LDL-C and the management of hypercholesterolemia in treatment.
- It is predicted that the hospital pharmacies segment will have a share of 38% by 2035, supported by increased dependency on the hospital-based dispensing of patients with specialized cardiovascular and hyperlipidemia care.
- As of the projections, the Asia Pacific will witness the highest CAGR of any region during the forecast period due to the rapidly growing healthcare infrastructure, increasing burden of lifestyle diseases, and the increasing accessibility of patients to lipid-lowering therapy.
Significant Growth Factors
The Bile Acid Sequestrants Market Trends present significant growth opportunities due to several factors:
- Rising Prevalence of Hypercholesterolemia and Cardiovascular Diseases: The growing international burden of hypercholesterolemia and related cardiovascular morbidity is the leading impetus behind the demand for bile acid sequestrants. In 2017-2020, 10% of all adults aged 20 years and above across the world have total cases of hyperlipidemia of more than 240 mg/dL, and about 17% have less than 40 mg/dL of high-density lipoprotein (HDL, or good) cholesterol. Cardiovascular disease (CVD) is the number one cause of death worldwide with an estimated 17.9 million deaths each year according to the world health organisation as a result of cardiovascular disease, which is 32% of all deaths worldwide. One of the most modifiable risk factors of atherosclerotic cardiovascular disease (ASCVD) is the presence of high LDL cholesterol, which is the main target of bile acid sequestrant therapy and poses a long-term clinical need for effective lipid-lowering pharmacotherapy. The market for bile acid sequestrants is experiencing tremendous growth due to rising incidences of hypercholesterolemia and other lipid disorders across the globe. These drugs act by attaching themselves to the bile acids in the gastrointestinal tract, preventing their reabsorption and triggering the liver to produce more bile acids out of the circulating cholesterol and thus decreasing the serum LDL-C. The LDL-C reductions with bile acid sequestrants have been found to be 10% to 30% when used as monotherapy and this varies with the dosage used. The growing patient population consists of not only adults with primary hypercholesterolemia but also those with familial hypercholesterolemia (FH), statin intolerant patients needing alternative lipid management, children with heterozygous FH and those with type 2 diabetes who need to have both their lipid and glucose control maintained. Market demand is further propelled by the growing geriatric population, which is a highly vulnerable population to hyperlipidemia. The elderly demographic aged 65 and over is expected to reach more than 1.5 billion patients around the world by 2050, which is an enormous and continuous group of patients that demand lipid-lowering agents such as bile acid sequestrants.
- Expanding Clinical Indications and Combination Therapy Adoption: through the extension of clinical indications of bile acid sequestrants beyond primary hypercholesterolemia, growth has increased significantly. Bile acid sequestrants have been approved by the FDA to treat hypercholesterolemia and may be combined with HMG-CoA reductase inhibitors (statins) or used as monotherapy. They also assist in the treatment of pruritus caused by high levels of bile acids and as adjunctive treatment for patients with hyperthyroidism in Graves’ disease. The FDA approval of Colesevelam to enhance glycemic control in adult patients with type 2 diabetes has been market-expanding especially since T2DM is currently affecting more than 537 million adults around the world with a forecast of over 783 million by the year 2045. This twofold signifies that colesevelam is one therapeutic agent that can be used to treat both dysglycemia and dyslipidemia in patients with comorbid T2DM and hypercholesterolemia – another common clinical combination. According to the 2018 AHA/ACC guidelines, cholesterol absorption inhibitors, bile acid sequestrants, and PCSK9 inhibitors should be considered in patients who do not respond to statin monotherapy with therapeutic results. The recommendation of bile acid sequestrants as second or third-line therapy is the most effective way to guarantee the persistence of prescription activity in cardiology and primary care environments worldwide. Combination regimens with bile acid sequestrants and statins, ezetimibe, or PCSK9 inhibitors are becoming increasingly used in the management of high-risk patients, which has a synergistic effect on the dynamics of demand.
What are the Major Advances Changing the Bile Acid Sequestrants Market Today?
- Next-Generation Formulations with Improved Tolerability: One of the main issues limiting the wider use of bile acid sequestrants has traditionally been the adverse effects profile of the gastrointestinal tract of the first-generation agents. Among the most general side effects are gastrointestinal which include constipation, stomach ache, bloating, vomiting, heartburn, loss of appetite, nausea and upset stomach. There is constipation experienced by one in ten patients taking colestipol and one in twenty-eight patients taking cholestyramine and they mostly occur with higher doses and with older patients aged above 65 years. Pharmaceutical developers have in turn responded by investing heavily in next-generation formulation strategies to enhance patient tolerability and medication adherence. Colesevelam is a polymer that was created as a more specific anion-binding reagent to overcome the gastrointestinal adverse effect and drug interactions of the previous nonspecific higher-volume bile acid binding resins. The advent of colesevelam tablets and oral suspension formulations has significantly enhanced the patient experience in comparison to its predecessors, in powder formulations, as the adherence rate has also increased, especially in elderly patients with difficulty in taking large amounts of powder.
- Colesevelam’s Dual Indication Driving Premium Market Segment Growth: The unique pharmacological properties of colesevelam as a lipid-lowering and glucose-lowering drug are fueling the premium segment in the bile acid sequestrant market. Colesevelam hydrochloride in three randomized, placebo-controlled studies involving patients with type 2 diabetes demonstrated a decrease in LDL-C and hemoglobin A1c, which prompted the FDA to approve colesevelam as an adjuvant to exercise and dietary restriction to achieve glycemic control in patients with T2D. This differentiation enables colesevelam to have a formulary opportunity and prescription market share in both cardiovascular and endocrinology therapeutic segments, establishing two physician prescriber channels cardiologists and endocrinologists which enhance commercial penetration. Colesevelam has the potential to treat two conditions simultaneously, which in the case of cardiometabolic patients’ management, where hyperlipidemia and T2DM often co-occur, alleviates the burden of polypharmacy and streamlines treatment processes. Incremental changes of 0.3 to 0.5% in glycemic control seen in clinical trials, although somewhat small, are clinically significant in patients who are already receiving background antidiabetic therapy. The T2DM signal has been especially significant in maintaining the revenue stream since cholestyramine and colestipol have been subjected to a lot of generic pressure that has removed the branded price strength.
- Pediatric Indications and Familial Hypercholesterolemia Management: Bile acid sequestrants have a clinically distinct role in the management of lipids in children. The FDA has also approved some bile acid sequestrants as safe in adolescents (10 to 17 years old), and bile acid sequestrants are among the limited drugs that are believed to be safe in children with familial hypercholesterolemia. Familial hypercholesterolemia is found in around 1 in 250 people worldwide, the more common type, heterozygous FH, leads to LDL-C levels 2 to 3 times higher than normal since birth, and this causes cardiovascular risk to be very high in the untreated condition throughout their lives. The cardiology guidelines are becoming increasingly supportive of early pharmacologic intervention of pediatric FH patients, and the American Academy of Pediatrics suggests the introduction of statins as early as age 810 in high-risk children. In children and adolescents in whom the initiation of statin therapy can be delayed or integrated into the adjunct therapy regimen, the existing pediatric safety and efficacy profile of colesevelam can be used in the initial treatment of such patients. The increase in awareness of early cardiovascular risk screening programs is increasing the population of the diagnosed pediatric patients, which is translated into the growing need for age-appropriate bile acid sequestrant formulations.
- Digital Health Integration and Medication Adherence Technologies: Digital health platforms, telemedicine, and remote patient monitoring of chronic diseases are transforming the prescribing and adherence patterns of bile acid sequestrants. Cardiovascular disease applications, linked pharmacy, and physician-patient interaction software are enhancing long-term medication adherence in the management of hypercholesterolemia – traditionally a major issue due to the lack of symptoms of high cholesterol. The current pharmacy segment of the hospital holds a huge market share, but the retail and online pharmacy channels are likely to experience huge growth with more access and convenience. Through telehealth platforms, the primary care physicians and cardiologists are able to remotely check lipid levels, modify dosages, and advise patients on the importance of compliance and changing their diets without necessarily having to visit them in-person, especially the elderly and patients with limited mobility. E-prescribing systems and electronic medication management portals are facilitating the prescribing process of bile acid sequestrants as adjunct therapy and minimizing administrative barriers that slowed adoption in the past. Pharmaceutical firms are now turning to digital patient support programs that accompany their branded bile acid sequestrant products that offer medication reminders and dietary advice and virtual nurse coaching to help patients stick with their treatment and decrease the rates of treatment discontinuation.
Category Wise Insights
By Drug Type
Why Does Colestipol Lead the Drug Type Segment?
Colestipol segment is expected to have a 35% market share of all the bile acid sequestrant markets in the world by the end of 2035, and this is expected to grow due to its wide application in the lowering of LDL cholesterol. Colestipol is gaining popularity as a treatment for primary hypercholesterolemia. It is introduced as an adjunctive intervention to dietary interventions and exercise. As for the Abolition of LDL-C levels, a 12 to 24% reduction is associated with doses of 4 to 16 grams per day. It can also reduce the chances of coronary artery disease. Colestipol is used off-label to treat cholestatic pruritus and irritable bowel syndrome or bile acid diarrhea.
Cholestyramine and colestipol are bile acid binding resins that have been used in clinical practice since the 1960s and come in a number of different formulations as generics. The accessibility of generic formulations at low prices has greatly increased the use of colestipol in cost-sensitive healthcare environments as well as in patients subjected to any type of public reimbursement plans with a preference to use generic formulations. The two formulations of colestipol, as tablets and in powder formulation, fit the different preferences of the patient, since patients who have difficulty with taking the powder formulation can use the tablet form, and patients who have a low tolerance to the pill burden may use the flavoured powder formulations. Colesevelam comes in 625 mg tablet or oral suspension packets in 1 or 2 divided doses, with the typical adult dose of 3.75 g/day. The FDA has recently passed a chewable bar type of colesevelam which is meant to be used together with meals. This ongoing development innovation by the manufacturer of colesevelam will likely help it in maintaining a premium segment price and brand loyalty in spite of the generic competition in the larger market.
Cholestyramine is the most prescribed bile acid sequestrant, as it has decades of experience and has a well-defined safety profile, whereas colesevelam is becoming popular because of its positive tolerability profile and potential cardiovascular effects other than lowering lipids. The historical LRC-CPPT clinical trial indicated that cholestyramine 24 g/day decreased LDL-C by about 23.5 mg/dL and was linked with a subsequent 19% decrease in the combined risk of coronary heart disease death and non-fatal myocardial infarction which forms the basis of the clinical acceptance of the drug class. Although this cardiovascular outcome data was produced using first-generation agents, it still supports the guideline inclusion of the entire drug category.
By Distribution Channel
Why Do Hospital Pharmacies Lead Distribution?
It will be projected that the bile acid sequestrant market in the hospital pharmacies segment will have the highest share of 38% by the year 2035. The rationale of such a large expansion of this segment is that once a patient suffers a huge heart attack and needs more specific care and treatment, the hospital pharmacies become the main provider of the medications to such individuals. Hospital pharmacies are the key initial contact of patients with severe cases of hypercholesterolemia or familial hypercholesterolemia or patients hospitalized due to acute cardiovascular events who are eventually placed on multifaceted lipid-lowering protocols involving combination therapy.
There will be significant growth in the retail and online pharmacy channels as more people will be able to access it and it will be more convenient. The shift of long-term, steady patients with hypercholesterolemia requiring hospital-based management to community management by retail pharmacies is not a new phenomenon, and retail pharmacies are progressively becoming the steady-state dispensing vehicle in patients on continuous lipid-lowering therapy on maintenance. The growth of retail pharmacy chains in the emerging markets, especially in Southeast Asia, Brazil, and the Middle East, is expanding access to bile acid sequestrants in geographies traditionally underserved by hospital dispensing. The online pharmacy is the most rapidly expanding distribution channel due to the use of digital prescriptions, direct to consumer chronic disease management websites, and ease of home delivery among chronic drug users. The available data on patient adherence indicate that online pharmacies with auto-refill and home delivery services have a significant effect on reducing the rates of therapy discontinuation in asymptomatic chronic diseases such as hypercholesterolemia.
Report Scope
| Feature of the Report | Details |
| Market Size in 2026 | USD 23.31 billion |
| Projected Market Size in 2035 | USD 45.77 billion |
| Market Size in 2025 | USD 21.8 billion |
| CAGR Growth Rate | 7.7% CAGR |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Key Segment | By Drug Type, Indication, Distribution Channel and Region |
| Report Coverage | Revenue Estimation and Forecast, Company Profile, Competitive Landscape, Growth Factors and Recent Trends |
| Regional Scope | North America, Europe, Asia Pacific, Middle East & Africa, and South & Central America |
| Buying Options | Request tailored purchasing options to fulfil your requirements for research. |
Regional Analysis
How Big is the North America Bile Acid Sequestrants Market?
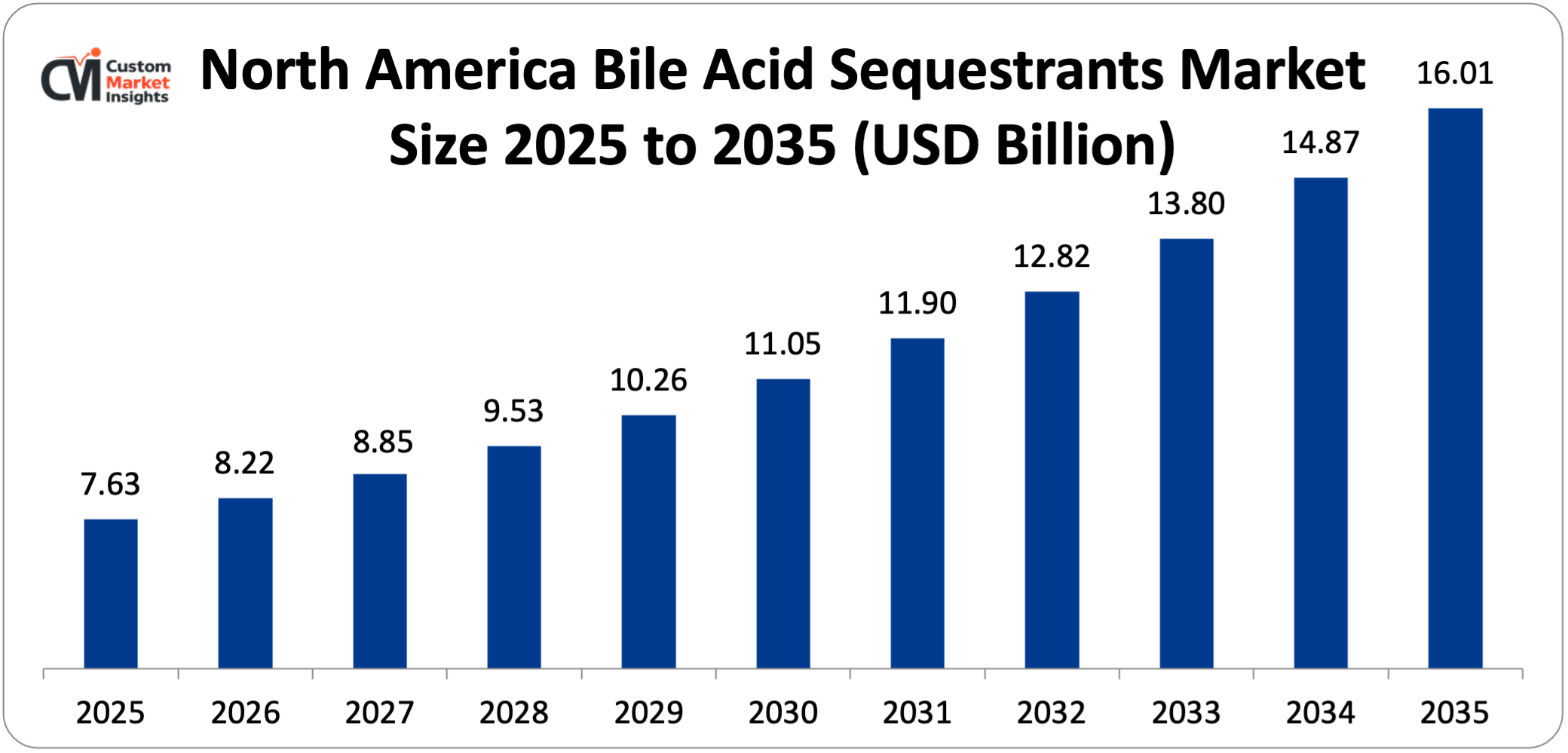
North America dominates the BAS market, accounting for over 35% of the global market share. The North American bile acid sequestrants market is estimated at approximately USD 7.63 billion in 2025 and is projected to reach USD 16.01 billion by 2035, expanding at a CAGR of 7.7% from 2026 to 2035.
Why does North America Lead the Bile Acid Sequestrants Market?
The bile acid sequestrants market is dominated by North America because of the high presence of large pharmaceutical firms, well developed healthcare facilities and high awareness of cholesterol control. The United States, specifically, boasts some of the key industry players that are undertaking research and development in order to improve drug formulations and increase the therapeutic uses. Combination therapy with statins is also gaining momentum, which contributes to the high demand of bile acid sequestrants in the region due to the high prevalence of hypercholesterolemia and cardiovascular diseases in the region.
The extensive coverage of the bile acid sequestrant therapy by the U.S. healthcare system through Medicare Part D, Medicaid, and private insurance guarantees the constant access of patients to the therapy, and colesevelam in particular can be strengthened by its formulary inclusion in the managed care plans that cover cardiovascular and diabetes treatments. The positive recommendation by the American Heart Association and the American College of Cardiology remains valid to approve the use of bile acid sequestrants as an adjunctive treatment in patients who failed to achieve LDL-C goals on statins and ezetimibe monotherapy.
U.S. Bile Acid Sequestrants Market Trends
The U.S. market is advantaged by a well-established prescribing culture among cardiologists and primary care physicians that fully acknowledges the decades-long history of safety of bile acid sequestrants, increased uptake of colesevelam in endocrinology practices as a T2DM glycemic control agent, and the continued expansion of the generic cholestyramine and colestipol market that further increases affordable Guidelines in cardiovascular disease prevention. Guidelines are still moving to more aggressive LDL-C levels, especially in very-high-risk patients, which presents clinical situations in which add-on treatment with bile acid sequestrants would be necessary to meet guideline-recommended levels. The existence of large pharmaceutical manufacturers and CDMOs that contribute to the reliability of supply chains only increases the dominant position of the U.S. market.
Why is Asia Pacific Experiencing the Fastest Growth?
The Asia Pacific will experience the most rapid growth during the forecast period, which is mainly given by the rising occurrence of lifestyle diseases, rise in disposable income, and healthcare expenditure. The Asia Pacific market of the bile acid sequestrants is expected to grow at a CAGR of about 9.4% between 2026 and 2035, the highest of all regions, due to the colossal magnitude of cardiovascular risk in the region. China and India carry almost half of the world’s diabetes burden, and the Asian phenotype disposition to dyslipidemia and cardiometabolic disease at lower BMI cutoffs presents a very high patient population that can be addressed with lipid-lowering drugs. Cholesterol screening penetration is being propelled by government-led non-communicable disease prevention programs across China, India, Japan, South Korea, and Southeast Asia, which increases the number of patients diagnosed that can be given pharmacotherapy.
China Bile Acid Sequestrants Market Trends
The Healthy China 2030 initiative of healthcare reforms in China and the expansion of the National Reimbursement Drug List (NRDL) to cover a wider variety of lipid-lowering agents are enhancing patients’ access to bile acid sequestrants. The ageing population in China, where the number of individuals aged 60 and above is more than 280 million, is an enormous demographic force contributing to the demand of cardiovascular medication. The generic pharmaceutical manufacturers in the country are increasingly producing cheaper colestipol and cholestyramine preparations which extends affordability in tier-2 and tier-3 cities.
Why is Europe Entering a New Era of Growth?
It is expected that Europe will witness significant expansion by the years 2026-2035 due to the increase in the prevalence of diabetes and the need to get more elaborate hyperlipidemia treatment. The market of European bile acid sequestrants is well-favored by national health system reimbursement systems developed in Germany, France, the United Kingdom, and Nordic countries to offer extensive coverage of lipid-lowering pharmacotherapy. The European Society of Cardiology (ESC) has issued European cardiology guidelines that support the use of bile acid sequestrants as an adjunct lipid-lowering agent in patients who do not reach LDL-C goals during statin maximization, favoring a uniform model of bile acid sequestrants across member states. The European market is dominated by countries such as Germany and the UK with regards to healthcare spending and cardiovascular management innovation. The statutory health insurance (GKV) system of Germany is covering more than 90% of the population and includes a full reimbursement of cholesterol-lowering medications, which supports the steady prescription volumes.
Germany Bile Acid Sequestrants Market Trends
Germany has the largest national market in Europe for bile acid sequestrants due to a robust and well-established system of pharmaceutical reimbursements, a significant and fast-aging population with high cardiovascular disease penetration and a well-developed network of hospital and ambulatory cardiology services to offer intensive lipid management services. Physicians in Germany show high compliance with ESC guidelines and advocate systematic use of add-on lipid-lowering agents such as bile acid sequestrants in high-risk patients.
Why is LAMEA Showing Accelerating Adoption?
The LAMEA region is characterized by slowly growing market development, where the growth is caused by the fact that healthcare infrastructure in the countries of the Middle East Gulf Cooperation Council improves, that the population in the urbanizing world becomes more aware of the cardiovascular risk, and that the pharmaceutical distribution networks in Brazil and South Africa are expanding. The Asia-Pacific and Latin American emerging economies offer growth potential, and the increasing disposable incomes and increased infrastructure for healthcare favour gradual market penetration. The cardiovascular disease burden in Brazil (the nation has more than 400,000 cardiovascular mortalities every year) and the large public health system in Brazil (SUS) are emerging platforms to further penetrate with lipid-lowering therapy including bile acid sequestrants.
Brazil Bile Acid Sequestrants Market Trends
The market expansion in Brazil is facilitated by the growing coverage of the public and private health insurance programs, the greater awareness of cardiologists and general practitioners about the comprehensive lipid management regimes, and the development of the generic pharmaceutical production industry that allows the supply of drugs at affordable prices in the country. The Brazilian government efforts to prevent and treat dyslipidemia in the rapidly urbanizing population through cardiovascular health programs such as early screening and treatment provide a good opportunity to support the long-term demand.
Top Players in the Bile Acid Sequestrants Market and Their Offerings
- Pfizer Inc.
- AstraZeneca Plc
- Novartis International AG
- Sanofi S.A.
- Amgen Inc.
- Merck & Co. Inc.
- AbbVie Inc.
- GlaxoSmithKline plc.
- Daiichi Sankyo Company Limited
- Eisai Co. Ltd.
- Abbott Laboratories
- Sun Pharmaceuticals
- Reddy’s Laboratories
- Astellas Pharma Inc.
- Others
Key Developments
The bile acid sequestrants market has experienced massive developments in the last two years with the industry players trying to increase the market capabilities and improve product holdings.
- At the beginning of 2025, one of the major pharmaceutical firms released a press statement indicating the commencement of a Phase III clinical trial of a novel polymer based bile acid sequestrant formulation that is capable of providing the same LDL-C lowering efficacy as the current agents but at a much reduced daily pill burden and with a much improved gastrointestinal tolerability profile.
- In late 2024, a large generics company was granted regulatory clearance in several Asian markets of a tablet formulation of cholestyramine with an optimized excipient profile to increase palatability and patient compliance compared to traditional powder formulations to capitalize on the emerging population of cardiovascular disease patients in China, South Korea, and Southeast Asia.
These strategic initiatives have enabled firms to gain market presence strength, broaden therapeutic accessibility, enhance patient compliance and adherence, and exploit the growth prospects in the growing international market of bile acid sequestrants.
The Bile Acid Sequestrants Market is segmented as follows:
By Drug Type
- Cholestyramine (Prevalite, Questran, Questran Lite)
- Colesevelam (Welchol)
- Colestipol (Colestid, Flavored Colestid)
- Others
By Indication
- Hypercholesterolemia
- Primary Hypercholesterolemia
- Familial Hypercholesterolemia (Heterozygous)
- Mixed Dyslipidemia
- Type 2 Diabetes Mellitus (Glycemic Control Adjunct)
- Cholestasis and Biliary Obstruction (Pruritus Relief)
- Bile Acid Malabsorption and Diarrhea
- Others
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Drug Stores
- Others
Regional Coverage:
North America
- U.S.
- Canada
- Mexico
- Rest of North America
Europe
- Germany
- France
- U.K.
- Russia
- Italy
- Spain
- Netherlands
- Rest of Europe
Asia Pacific
- China
- Japan
- India
- New Zealand
- Australia
- South Korea
- Taiwan
- Rest of Asia Pacific
The Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of the Middle East & Africa
Latin America
- Brazil
- Argentina
- Rest of Latin America
Table of Contents
- Chapter 1. Report Introduction
- 1.1. Report Description
- 1.1.1. Purpose of the Report
- 1.1.2. USP & Key Offerings
- 1.2. Key Benefits For Stakeholders
- 1.3. Target Audience
- 1.4. Report Scope
- 1.1. Report Description
- Chapter 2. Market Overview
- 2.1. Report Scope (Segments And Key Players)
- 2.1.1. Bile Acid Sequestrants by Segments
- 2.1.2. Bile Acid Sequestrants by Region
- 2.2. Executive Summary
- 2.2.1. Market Size & Forecast
- 2.2.2. Bile Acid Sequestrants Market Attractiveness Analysis, By Drug Type
- 2.2.3. Bile Acid Sequestrants Market Attractiveness Analysis, By Indication
- 2.2.4. Bile Acid Sequestrants Market Attractiveness Analysis, By Distribution Channel
- 2.1. Report Scope (Segments And Key Players)
- Chapter 3. Market Dynamics (DRO)
- 3.1. Market Drivers
- 3.1.1. Rising Prevalence of Hypercholesterolemia and Cardiovascular Diseases
- 3.1.2. Expanding Clinical Indications and Combination Therapy Adoption
- 3.2. Market Restraints
- 3.3. Market Opportunities
- 3.5. Pestle Analysis
- 3.6. Porter Forces Analysis
- 3.7. Technology Roadmap
- 3.8. Value Chain Analysis
- 3.9. Government Policy Impact Analysis
- 3.10. Pricing Analysis
- 3.1. Market Drivers
- Chapter 4. Bile Acid Sequestrants Market – By Drug Type
- 4.1. Drug Type Market Overview, By Drug Type Segment
- 4.1.1. Bile Acid Sequestrants Market Revenue Share, By Drug Type, 2025 & 2035
- 4.1.2. Cholestyramine (Prevalite, Questran, Questran Lite)
- 4.1.3. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 4.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.5. Key Market Trends, Growth Factors, & Opportunities
- 4.1.6. Colesevelam (Welchol)
- 4.1.7. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 4.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.9. Key Market Trends, Growth Factors, & Opportunities
- 4.1.10. Colestipol (Colestid, Flavored Colestid)
- 4.1.11. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 4.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.13. Key Market Trends, Growth Factors, & Opportunities
- 4.1.14. Others
- 4.1.15. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 4.1.16. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.17. Key Market Trends, Growth Factors, & Opportunities
- 4.1. Drug Type Market Overview, By Drug Type Segment
- Chapter 5. Bile Acid Sequestrants Market – By Indication
- 5.1. Indication Market Overview, By Indication Segment
- 5.1.1. Bile Acid Sequestrants Market Revenue Share, By Indication, 2025 & 2035
- 5.1.2. Hypercholesterolemia
- 5.1.2.1. Primary Hypercholesterolemia
- 5.1.2.2. Familial Hypercholesterolemia (Heterozygous)
- 5.1.2.3. Mixed Dyslipidemia
- 5.1.3. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 5.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.5. Key Market Trends, Growth Factors, & Opportunities
- 5.1.6. Type 2 Diabetes Mellitus (Glycemic Control Adjunct)
- 5.1.7. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 5.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.9. Key Market Trends, Growth Factors, & Opportunities
- 5.1.10. Cholestasis and Biliary Obstruction (Pruritus Relief)
- 5.1.11. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 5.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.13. Key Market Trends, Growth Factors, & Opportunities
- 5.1.14. Bile Acid Malabsorption and Diarrhea
- 5.1.15. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 5.1.16. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.17. Key Market Trends, Growth Factors, & Opportunities
- 5.1.18. Others
- 5.1.19. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 5.1.20. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.21. Key Market Trends, Growth Factors, & Opportunities
- 5.1. Indication Market Overview, By Indication Segment
- Chapter 6. Bile Acid Sequestrants Market – By Distribution Channel
- 6.1. Distribution Channel Market Overview, By Distribution Channel Segment
- 6.1.1. Bile Acid Sequestrants Market Revenue Share, By Distribution Channel, 2025 & 2035
- 6.1.2. Hospital Pharmacies
- 6.1.3. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 6.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.5. Key Market Trends, Growth Factors, & Opportunities
- 6.1.6. Retail Pharmacies
- 6.1.7. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 6.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.9. Key Market Trends, Growth Factors, & Opportunities
- 6.1.10. Online Drug Stores
- 6.1.11. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 6.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.13. Key Market Trends, Growth Factors, & Opportunities
- 6.1.14. Others
- 6.1.15. Bile Acid Sequestrants Share Forecast, By Region (USD Billion)
- 6.1.16. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.17. Key Market Trends, Growth Factors, & Opportunities
- 6.1. Distribution Channel Market Overview, By Distribution Channel Segment
- Chapter 7. Bile Acid Sequestrants Market – Regional Analysis
- 7.1. Bile Acid Sequestrants Market Overview, By Region Segment
- 7.1.1. Global Bile Acid Sequestrants Market Revenue Share, By Region, 2025 & 2035
- 7.1.2. Global Bile Acid Sequestrants Market Revenue, By Region, 2026 – 2035 (USD Billion)
- 7.1.3. Global Bile Acid Sequestrants Market Revenue, By Drug Type, 2026 – 2035
- 7.1.4. Global Bile Acid Sequestrants Market Revenue, By Indication, 2026 – 2035
- 7.1.5. Global Bile Acid Sequestrants Market Revenue, By Distribution Channel, 2026 – 2035
- 7.2. North America
- 7.2.1. North America Bile Acid Sequestrants Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 7.2.2. North America Bile Acid Sequestrants Market Revenue, By Drug Type, 2026 – 2035
- 7.2.3. North America Bile Acid Sequestrants Market Revenue, By Indication, 2026 – 2035
- 7.2.4. North America Bile Acid Sequestrants Market Revenue, By Distribution Channel, 2026 – 2035
- 7.2.5. U.S. Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.2.6. Canada Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.2.7. Mexico Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.2.8. Rest of North America Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.3. Europe
- 7.3.1. Europe Bile Acid Sequestrants Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 7.3.2. Europe Bile Acid Sequestrants Market Revenue, By Drug Type, 2026 – 2035
- 7.3.3. Europe Bile Acid Sequestrants Market Revenue, By Indication, 2026 – 2035
- 7.3.4. Europe Bile Acid Sequestrants Market Revenue, By Distribution Channel, 2026 – 2035
- 7.3.5. Germany Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.3.6. France Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.3.7. U.K. Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.3.8. Russia Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.3.9. Italy Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.3.10. Spain Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.3.11. Netherlands Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.3.12. Rest of Europe Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.4. Asia Pacific
- 7.4.1. Asia Pacific Bile Acid Sequestrants Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 7.4.2. Asia Pacific Bile Acid Sequestrants Market Revenue, By Drug Type, 2026 – 2035
- 7.4.3. Asia Pacific Bile Acid Sequestrants Market Revenue, By Indication, 2026 – 2035
- 7.4.4. Asia Pacific Bile Acid Sequestrants Market Revenue, By Distribution Channel, 2026 – 2035
- 7.4.5. China Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.4.6. Japan Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.4.7. India Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.4.8. New Zealand Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.4.9. Australia Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.4.10. South Korea Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.4.11. Taiwan Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.4.12. Rest of Asia Pacific Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.5. The Middle-East and Africa
- 7.5.1. The Middle-East and Africa Bile Acid Sequestrants Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 7.5.2. The Middle-East and Africa Bile Acid Sequestrants Market Revenue, By Drug Type, 2026 – 2035
- 7.5.3. The Middle-East and Africa Bile Acid Sequestrants Market Revenue, By Indication, 2026 – 2035
- 7.5.4. The Middle-East and Africa Bile Acid Sequestrants Market Revenue, By Distribution Channel, 2026 – 2035
- 7.5.5. Saudi Arabia Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.5.6. UAE Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.5.7. Egypt Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.5.8. Kuwait Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.5.9. South Africa Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.5.10. Rest of the Middle East & Africa Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.6. Latin America
- 7.6.1. Latin America Bile Acid Sequestrants Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 7.6.2. Latin America Bile Acid Sequestrants Market Revenue, By Drug Type, 2026 – 2035
- 7.6.3. Latin America Bile Acid Sequestrants Market Revenue, By Indication, 2026 – 2035
- 7.6.4. Latin America Bile Acid Sequestrants Market Revenue, By Distribution Channel, 2026 – 2035
- 7.6.5. Brazil Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.6.6. Argentina Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.6.7. Rest of Latin America Bile Acid Sequestrants Market Revenue, 2026 – 2035 (USD Billion)
- 7.1. Bile Acid Sequestrants Market Overview, By Region Segment
- Chapter 8. Competitive Landscape
- 8.1. Company Market Share Analysis – 2025
- 8.1.1. Global Bile Acid Sequestrants Market: Company Market Share, 2025
- 8.2. Global Bile Acid Sequestrants Market Company Market Share, 2024
- 8.1. Company Market Share Analysis – 2025
- Chapter 9. Company Profiles
- 9.1. Pfizer Inc.
- 9.1.1. Company Overview
- 9.1.2. Key Executives
- 9.1.3. Product Portfolio
- 9.1.4. Financial Overview
- 9.1.5. Operating Business Segments
- 9.1.6. Business Performance
- 9.1.7. Recent Developments
- 9.2. AstraZeneca Plc
- 9.3. Novartis International AG
- 9.4. Sanofi S.A.
- 9.5. Amgen Inc.
- 9.6. Merck & Co. Inc.
- 9.7. AbbVie Inc.
- 9.8. GlaxoSmithKline plc.
- 9.9. Daiichi Sankyo Company Limited
- 9.10. Eisai Co. Ltd.
- 9.11. Abbott Laboratories
- 9.12. Sun Pharmaceuticals
- 9.13. Dr. Reddy’s Laboratories
- 9.14. Astellas Pharma Inc.
- 9.15. Others.
- 9.1. Pfizer Inc.
- Chapter 10. Research Methodology
- 10.1. Research Methodology
- 10.2. Secondary Research
- 10.3. Primary Research
- 10.3.1. Analyst Tools and Models
- 10.4. Research Limitations
- 10.5. Assumptions
- 10.6. Insights From Primary Respondents
- 10.7. Why Healthcare Foresights
- Chapter 11. Standard Report Commercials & Add-Ons
- 11.1. Customization Options
- 11.2. Subscription Module For Market Research Reports
- 11.3. Client Testimonials
- Chapter 12. List Of Figures
- 12.1. Figures No 1 to 32
- Chapter 13. List Of Tables
- 13.1. Tables No 1 to 46
Prominent Player
- Pfizer Inc.
- AstraZeneca Plc
- Novartis International AG
- Sanofi S.A.
- Amgen Inc.
- Merck & Co. Inc.
- AbbVie Inc.
- GlaxoSmithKline plc.
- Daiichi Sankyo Company Limited
- Eisai Co. Ltd.
- Abbott Laboratories
- Sun Pharmaceuticals
- Reddy’s Laboratories
- Astellas Pharma Inc.
- Others
FAQs
The key players in the market are Pfizer Inc., AstraZeneca Plc, Novartis International AG, Sanofi S.A., Amgen Inc., Merck & Co. Inc., AbbVie Inc., GlaxoSmithKline plc., Daiichi Sankyo Company Limited, Eisai Co. Ltd., Abbott Laboratories, Sun Pharmaceuticals, Dr. Reddy’s Laboratories, Astellas Pharma Inc., Others.
The cost of treatment of the branded formulations is high, there is the possibility of side effects due to the use of bile acid sequestrants, especially gastrointestinal effects, and the existence of alternative cholesterol-lowering treatments such as PCSK9 inhibitors and novel RNA-based therapeutics limit the growth of the market. Market expansion can also be affected by regulatory hurdles and high approval procedures on new drug development. The first-generation agents are experiencing intensified generic competition, and the introduction of highly effective novel lipid-lowering agents, such as inclisiran (siRNA therapy) and evolocumab/alirocumab (PCSK9 inhibitors), with superior LDL-C lowering efficacy in a small number of doses of injections, is posing the greatest competitive pressure in the high-end market segment.
It is estimated that the bile acid sequestrant market size in the world will reach USD 45.77 billion in revenue by 2035 with a CAGR of above 7.7%. This is strong growth that is indicative of the increasing prevalence of hypercholesterolemia, adoption of combination therapy as recommended by the guidelines, extension of clinical indications, patient accessibility in new markets, and continuous formulation innovation enhancing tolerance and compliance.
It is estimated that North America will take the largest portion of the bile acid sequestrants market by 2035 following the prevalence of cholestyramine in the treatment of pruritus symptoms among liver cirrhosis patients. The leading presence is strengthened with the existence of advanced healthcare infrastructure, full coverage of pharmaceutical reimbursement, a strong culture of prescription guided by guidelines, the presence of major pharmaceutical manufacturing firms, and high prevalence of cardiovascular diseases in the adult population of the U.S.
The region of Asia Pacific is expected to experience the highest CAGR in the forecast period at about 9.4% due to the massive and quickly increasing cardiometabolic patient populations in China and India, the rapidly aging populations across Japan and South Korea, the growing healthcare infrastructure and cholesterol screening rates, and the growing disposable incomes that will allow more individuals to access pharmaceuticals and the government initiatives to prevent non-communicable diseases.
The market of bile acid sequestrants is influenced by the increasing rates of hypercholesterolemia and cardiovascular diseases, the increase in the use of sedentary lifestyles and unhealthy diets, and the awareness of the role of lipid control and cardiovascular risk prevention. Other reasons are the guideline support of bile acid sequestrants as add-on therapy in patients who fail to achieve LDL-C goals with statins, the dual indication of colesevelam in T2DM glycemic control, the growing use of colesevelam in the management of children with familial hypercholesterolemia, and the increasing coverage of reimbursement in the emerging markets in the Asian Pacific. All these contribute to the global projection of a CAGR of 7.7% throughout 2026 to 2035.