Malaria Diagnostics Market Size, Trends and Insights By Technology (Rapid Diagnostic Tests (RDTs), Single Species Detection, Multi-Species Detection, Microscopy, Light Microscopy, Fluorescence Microscopy, Molecular Diagnostic Tests, Conventional PCR, Real-Time PCR, LAMP (Loop-Mediated Isothermal Amplification), Other Technologies), By Product Type (Diagnostic Kits & Reagents, Instruments, Software & Services), By End-User (Hospitals & Clinics, Diagnostic Centers, Academic & Research Institutes, Others), and By Region - Global Industry Overview, Statistical Data, Competitive Analysis, Share, Outlook, and Forecast 2026 – 2035
Report Snapshot
| Study Period: | 2026-2035 |
| Fastest Growing Market: | Asia Pacific |
| Largest Market: | Middle East & Africa |
Major Players
- Abbott Laboratories
- Access Bio Inc.
- Beckman Coulter Inc.
- Bio-Rad Laboratories Inc.
- Others
Reports Description
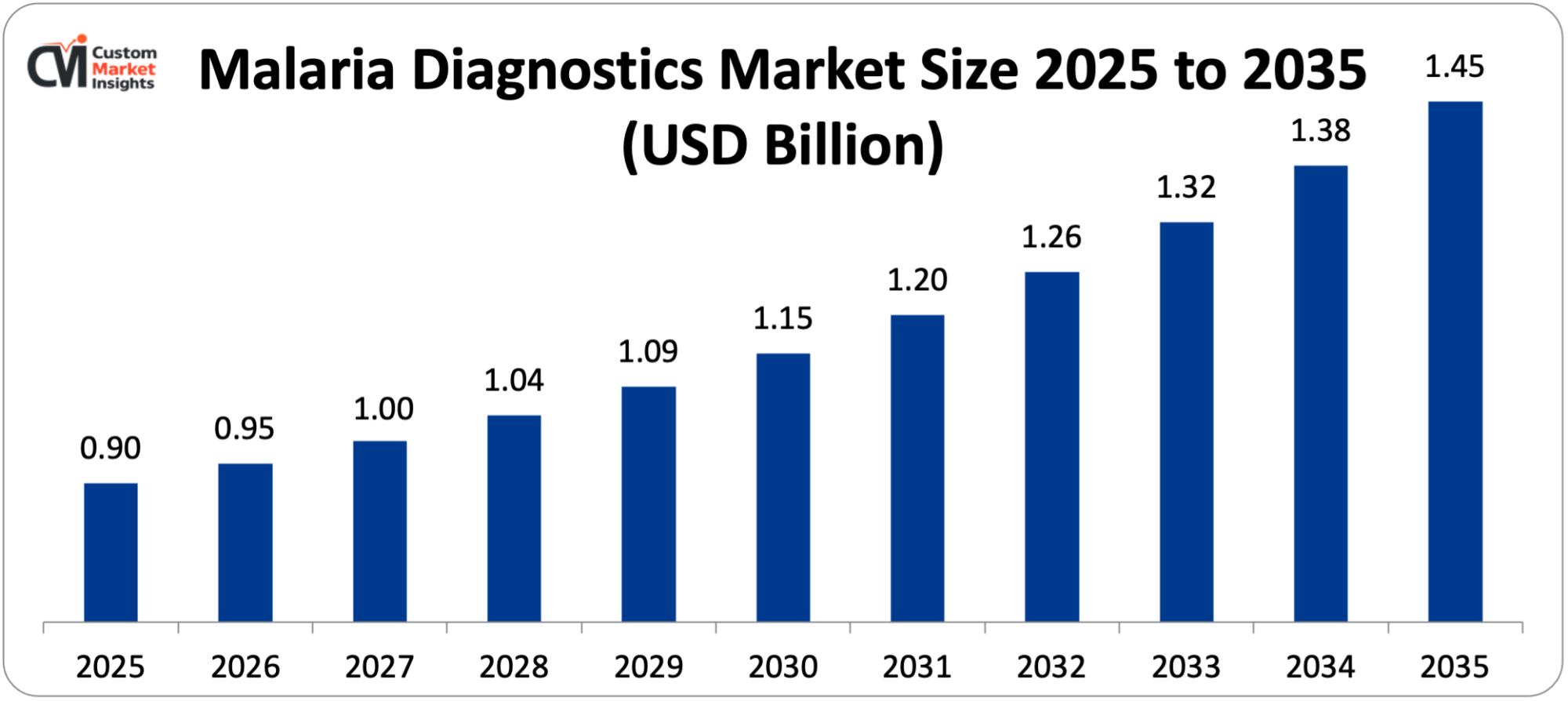
The malaria diagnostics market size in the world is estimated at USD 0.90 billion in 2025 and is projected to grow further by USD 0.95 billion in 2026 to about USD 1.45 billion by 2035 at a CAGR of 5.2% between 2026 and 2035. Market growth is contributed to by the growing prevalence of malaria in endemic areas, increased government and non-government efforts to eradicate malaria, the growing use of rapid diagnostic tests in the remote areas, and technological improvements in molecular diagnostic systems.
Market Highlight
- The Middle East & Africa as the suppliers of the malaria diagnostics market had a market share of 53% in 2025.
- Asia Pacific will grow by a CAGR of 7.8% during the period 2026 to 2035.
- Through technology, the rapid diagnostic tests segment had an estimated market share of approximately 54% by 2025.
- By technology, the segment of the molecular diagnostic tests is the fastest-growing with a CAGR of 8.4% between 2026 and 2035.
- End-user- The hospitals and clinics segment constituted the largest market share of 65% in the year 2025 with the diagnostic centers projected to have the highest CAGR of 6.8% in the years 2026 to 2035.
- By the type of products, diagnostic kits and reagents secured 66% of the market share in 2025.
Significant Growth Factors
Market Trends of the Malaria Diagnostics have tremendous growth prospects because of the following reasons:
- High Disease Burden in Endemic Regions and Global Elimination Initiatives: The main force of the diagnostics market is the present prevalence of malaria in tropical and subtropical countries as the disease continues to be among the deadliest infectious diseases in the world, despite decades of control measures. In 2020, data provided by the World Health Organization indicated that there were approximately 241 million malaria cases and 627,000 malaria-related deaths across the globe with 95% of all malaria cases and 96% of all malaria deaths said to occur in the African region. The disease targets the vulnerable in the society such as the children below the age of five years who constitute 80% of the deaths related to malaria in Africa, pregnant women who are at risk of severe illness and unfavorable prognoses and the poor communities who cannot access prevention and treatment. The prevalence of malaria in the world is as shown in the global distribution, of which 29 countries recorded 95% of the cases with six countries bearing about 55% of the global burden, comprising Nigeria (27%), the Democratic Republic of Congo (12%), Uganda (5%), Mozambique (4%), Angola (3.4%), and Burkina Faso (3.4%). The economic effect of malaria is not only limited to direct healthcare expenditure but the estimated USD 12 billion is the annual economic productivity lost due to absenteeism of the workforce, decreased productivity, and treatment costs. The global effort to eliminate malaria continues, with the World Health Organization introducing such initiatives as the Elimination 2025 Initiative, which targets 25 countries as potential elimination targets by 2025, and the End Malaria Faster strategy, which was announced in October 2021 by the U.S. Presidential Malaria Initiative as a measure to curb disparities and increase surveillance. Also, there is a significant influx of money to endemic nations, and Nigeria has received more than USD 2.6 billion in the form of the Global Fund to Fight AIDS, TB, and Malaria, and the United Kingdom has contributed about 206 billion shillings to Uganda in 5 years concentrating on vulnerable groups. Proper and available diagnostics are the foundations of successful malaria control programs, and they will help provide timely treatment and prevent the advancement of the disease, the formation of a carrier, and the development of resistance to drugs through the correct choice of therapy depending on the species. Contemporary diagnostic methods complement malaria surveillance systems by assisting in the detection of cases, tracking the trend of transmission, and assessing the intervention outcomes.
- Technological Advancements in Diagnostic Methods: Diagnostic technologies have highly enhanced the growth of the market through the advancement in the diagnostic accuracy, speed and availability of the diagnostic tests due to technological advancement in all testing modalities. Some of the innovations are the development of ultra-sensitive rapid diagnostic tests that are able to detect lower parasite densities than the previous generations, multiplex molecular assays that can detect more than one species of Plasmodium and can also detect co-infections; point-of-care molecular platforms that can provide PCR-level sensitivity in the field while not requiring a laboratory infrastructure and artificial intelligence-driven microscopy to automate the process of parasite detection and counting. In July 2022, Mylab Discovery Solutions released the PathoDetect Extended Monsoon Fever Panel, a malaria, dengue, chikungunya, Zika, Salmonellosis, Leptospirosis and Leishmaniasis multiplex RT-PCR kit showing integration in diagnostics across multiple endemic diseases. In October 2021, Sysmex presented the XN-30 RUO Automated Hematology Analyzer which was designed to detect malaria through violet laser technology and complex algorithms to identify parasitemia levels, which is an example of automation to minimize variability in human interpretation. Innovation in the alternative methodology of detection has been shown through the creation of Gazelle, an economical diagnostic tool of magneto-optical detection of hemozoin crystals created by all human malaria parasite species. In January 2019, ERADA Technology Alliance Ltd released SMAART, the first saliva-based rapid malaria detection test in the world that allows the use of non-invasive and asymptomatic screening to increase testing accessibility. In December 2019, with a joint effort of Access Bio and Global Good, ultra-sensitive rapid diagnostic tests were developed, which are five times more sensitive in detecting histidine-rich protein 2 (HRP2) antigens than the existing available tests, solving the problem of low-density parasitemia detection. Moreover, the combination with digital health platforms allows gathering data in real-time, conducting disease surveillance mapping, and tracking treatment, which enhances program management and resource allocation. Mobile-based diagnostic platforms which are the new complementary devices can be used to connect the field locations to the reference laboratories to provide the quality assurance and specialist consultation that will help in proper diagnosis in the remote locations.
What are the Major Advances Changing the Malaria Diagnostics Market Today
- Rapid Diagnostic Tests and Point-of-Care Testing: The most radical innovation in malaria diagnostics is the introduction of rapid diagnostic tests, the use of which allows the detection of the parasite with high accuracy in 15-20 minutes without the need to maintain laboratory facilities and specific training. The immunochromatographic method in RDTs is used to identify parasite-specific antigens such as the histidine-rich protein 2 (HRP2) produced by P. falciparum and plasmodium lactate dehydrogenase (pLDH) expressed by several Plasmodium species on test strips using monoclonal antibodies. The RDT segment represents 54% of the malaria diagnostics market in 2025 and is growing strongly as it is easy to use, requiring few training units, cost-effective with unit prices of USD 1 or less meaning it can be affordable in resource-constrained emergency conditions; fast, with results to make immediate treatment decisions, and portable enough to reach remote geographies where power is unavailable and there are no laboratories. As shown in the market data, single species detection tests prevail with a market share of 61.1%, whereas the multi-species tests are becoming more popular in the market to carry out overall diagnoses. Since 2011, the scale of RDT implementation in endemic areas is demonstrated by the fact that UNICEF alone purchases 10-11 million malaria rapid diagnostic tests every year. The U.S. government is an example of an international aid donating more than 1.7 million RDTs and 1.3 million malaria control treatment courses. RDTs allow decentralized testing that takes diagnostics to the village level to minimize time delays between symptom onset and treatment, which can lead to serious disease development and death.
- Molecular Diagnostics and Ultra-Sensitive Detection: Molecular diagnostic platforms such as polymerase chain reaction (PCR) and nucleic acid amplification tests are more sensitive and specific than standard methods to detect parasitemia down to 1-5 parasites per microliter rather than microscopy which has a limit of 50-100 parasites per microliter. Molecular diagnostics have the highest growth of 8.4% CAGR between 2026 and 2035, owing to their unparalleled level of sensitivity in low-density infection detection in elimination context, species differentiation that allows the selection of the appropriate treatment, the detection of resistance markers that identify mutations of drug resistance, and the reduction of inter-observer variability in microscopy. The contemporary molecular methods encompass real-time PCR technology of quantitative parasitemia, loop-mediated isothermal amplification (LAMP) of constant temperature, and cartridge-based technologies of sample preparation, amplification, and detection. The recent advances of point-of-care molecular platforms allow PCR-equivalent performance in the field, and such devices as the Cepheid GeneXpert system are currently used in remote clinics to give results within less than two hours. Molecular diagnostics find research uses such as tracking the development of drug resistance, epidemiology, and vaccination efficacy, in addition to routine clinical diagnosis.
- Artificial Intelligence and Automated Microscopy: This technology combines the use of artificial intelligence and conventional microscopy to overcome the long-standing limitations of operator competency requirements and inconsistencies in interpretations and still retains the benefits of microscopy such as species identification and quantitative parasitemia. The microscopy systems use AI to scan slides and automate the identification of parasite morphology, perform quantitative analysis to determine parasitemia, and produce high-resolution images of blood films, machine learning algorithms that are trained with thousands of annotated images, and quality control to flag slides that require a human microscopist. These systems provide stable performance even without the operator, they can also provide expert-level diagnosis at a facility with no experienced microscopists, and they can be improved by increasing throughput to allow processing of higher volumes of samples. Another method of automation is automated hematology analyzers such as the Sysmex XN-30 which identify malaria parasites as part of a routine complete blood count without necessarily having to perform specific tests to identify malaria. The technology has a specific potential in surveillance programs with high-throughput screening and elimination environments in which it is difficult to retain microscopist skills as the number of cases reduces.
- Multiplex and Co-Infection Diagnostic Panels: These panels are diagnostic tests that are designed to simultaneously identify multiple pathogens because it is a known fact that malaria endemic areas are frequently endemic to several different fever causing diseases with similar clinical presentations. Multiplex panels such as the PathoDetect Extended Monsoon Fever Panel identify malaria as well as dengue, chikungunya, Zika and bacterial infections and can be used to provide differential diagnosis to provide the appropriate treatment. The benefit of these multiplex strategies lies in their clinical use, wherein they result in accurate diagnosis, which leads to the reduction of inappropriate treatment; public health, where they are useful in the surveillance of a number of diseases simultaneously, and economical, where they allow the detection of several targets using a single test. The increasing awareness of the role of co-infections on disease severity and treatment results is a reason why the holistic diagnostic methods are adopted especially at a time when malaria patients can also be infected with the helminth infections, HIV, or bacterial co-infections that affect the manifestations of the disease and treatment outcomes.
Category Wise Insights
By Technology
Why Rapid Diagnostic Tests Dominate the Market?
The highest technology segment is in 2025 when there are rapid diagnostic tests which constitute around 54% of the total market share. This supremacy indicates how RDTs impact malaria diagnosis through transformation because they facilitate point-of-care testing in areas with scarce resources where the malaria burden is greatest. The principle of RDTs is the immunochromatographic technique to identify malaria parasite antigens in small quantities of blood, the outcome of which is observed after 15-20 minutes in color lines on test strips. The technology has been successful because of its simplicity, whereby training is minimal to deploy; cost-effectiveness, where unit prices usually do not exceed USD 1, hence large-scale deployment can be achieved, no equipment is required, so it can be used even without electricity or lab infrastructure and it can turn around results quickly thus treatments are made instantly.
RDTs have led to the democratization of malaria diagnosis because they have been able to bring testing facilities to the villages, informal settlements and conflict areas which previously had no access to testing. This decentralization will allow test-and-treat approaches in which patients can be diagnosed and treated during their initial healthcare visit, which has a tremendous effect on treatment delays that lead to serious disease outbreaks and death. The P. falciparum only single species RDTs represent 61.1% of the RDT market as P. falciparum is the dominant species and causes severe malaria that needs urgent treatment in Africa. In Asia and Latin America where P. vivax coexists with P. falciparum and thus requires species-specific treatment including primaquine to radically treat the former, multi-species RDTs with the ability to distinguish P. falciparum from the other Plasmodium species are gaining use.
Microscopy is a significant technology with about 35% of the market share, which is the conventional gold standard of diagnosing malaria. Microscopy allows identifying the species and conducting quantitative parasitemia evaluation, on the basis of which the treatment decision is made, as well as the possibility of identifying mixed infections involving two or more Plasmodium species. The technology involves the need to have trained microscopists, access to electricity to enhance the light, and periodic quality control, so it can be implemented only in health facilities that have the laboratory facility. These limitations notwithstanding, microscopy continues to have relevance in the confirmation of RDT-positive results, in the treatment failure cases, and in surveillance programs in which quantitative data of parasitemia shapes the interpretation of the intensity of transmission.
The highest growth is seen in molecular diagnostic tests with the CAGR of 8.4% between 2026 and 2035 due to the highest sensitivity of close to 1-5 parasites per microliter. Molecular platforms are superior in low-transmission and elimination conditions where identifying low-density infections (when they are asymptomatic) is crucial in preventing the residual transmission, monitoring drug resistance necessitating detection of resistance mutations, and research studies that require exact parasite quantification.
By Product Type
Why Diagnostic Kits & Reagents Dominate?
The highest product segment is diagnostic kits and reagents; this market share is about 66% in 2025. This has an impact of dominance on the consumable aspect of diagnostic testing, where a new test kit or reagent set is needed by individual patients so that the recurring revenue exceeds the investments in durable equipment. This segment contains various rapid diagnostic test kits with the representation of a single use disposable test, microscopy reagents such as Giemsa stain to prepare blood films, molecular diagnostic reagents such as PCR master mixes and primers, and quality control reagents to assure accuracy in the tests. The continuous consumption model establishes stable demand curves, and endemic regions are in need of millions of tests per year to feed the populations.
Notable technological advances such as increased sensitivity of later generations of RDTs, multiplexed methods of detecting multiple targets at once, and more shelf life in tropical environments have been seen in diagnostic kits. Corporations such as Abbott, Access Bio and Premier Medical Corporation are constantly developing to enhance their performance and cut costs, besides working on the new challenges facing the organization such as HRP2 gene deletions in P. falciparum leading to a decreased sensitivity on RDT.
Instruments are a minor yet significant category encompassing microscopes that can be used to test blood films, automated hematology devices that identify parasites, molecular diagnostic systems to test PCR, and quality control readers that test RDT performance. Although the sale of individual instruments is a great source of revenue, the installed base increases at a low rate as compared to the turnover of consumables.
By End-User
Why Hospitals & Clinics Lead Adoption?
The largest end-user market would be hospitals & clinics, which will claim about 65% of the market in 2025. This leadership is a representation of the focal position of formal healthcare facilities in the diagnosis and treatment of malaria as these facilities treat most of the diagnosed cases. Hospitals and clinics have a wide range of services that include diagnostic testing with RDTs, microscopy, and molecular testing, inpatient services for the severely malaria-related cases that need intensive management; outpatient services for the uncomplicated cases, and follow-up to ensure that treatment has been successful. Hospitals and clinics are the perfect place to diagnose malaria, as the skilled medical staff, laboratory facilities, and their connection with treatment services are concentrated there.
Endemic areas have hospitals, which contain high patient volumes and some of the hospitals in these areas identify hundreds of cases of malaria each day when the malaria epidemics are at their peak. These institutions enjoy international assistance such as donated diagnostic supplies, equipment and training programs that bolster diagnostic capacities. The expansion of the healthcare infrastructure in endemic nations contributes to the growth of the hospital segment, where new facilities need diagnostic devices and constant provision of consumables.
Diagnostic centers will be the quickest growing end-user segment with 6.8% CAGR between 2026 and 2035 due to the existence of specialized facilities that only offer diagnostic services. These centers normally use high-tech equipment such as automated analyzers and molecular platforms that offer high accuracy testing that appeals to patients who want a conclusive diagnosis. There is an increased development of diagnostic centers in urban centres of endemic nations and this increases accessibility to a greater number of populations wishing to access private health care. Their specialized services and special equipment provide them with high-quality diagnostic services to supplement the testing in the hospital.
Report Scope
| Feature of the Report | Details |
| Market Size in 2026 | USD 0.95 billion |
| Projected Market Size in 2035 | USD 1.45 billion |
| Market Size in 2025 | USD 0.90 billion |
| CAGR Growth Rate | 5.2% CAGR |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Key Segment | By Technology, Product Type, End-User and Region |
| Report Coverage | Revenue Estimation and Forecast, Company Profile, Competitive Landscape, Growth Factors and Recent Trends |
| Regional Scope | North America, Europe, Asia Pacific, Middle East & Africa, and South & Central America |
| Buying Options | Request tailored purchasing options to fulfil your requirements for research. |
Regional Analysis
How Big is the Middle East & Africa Market Size?
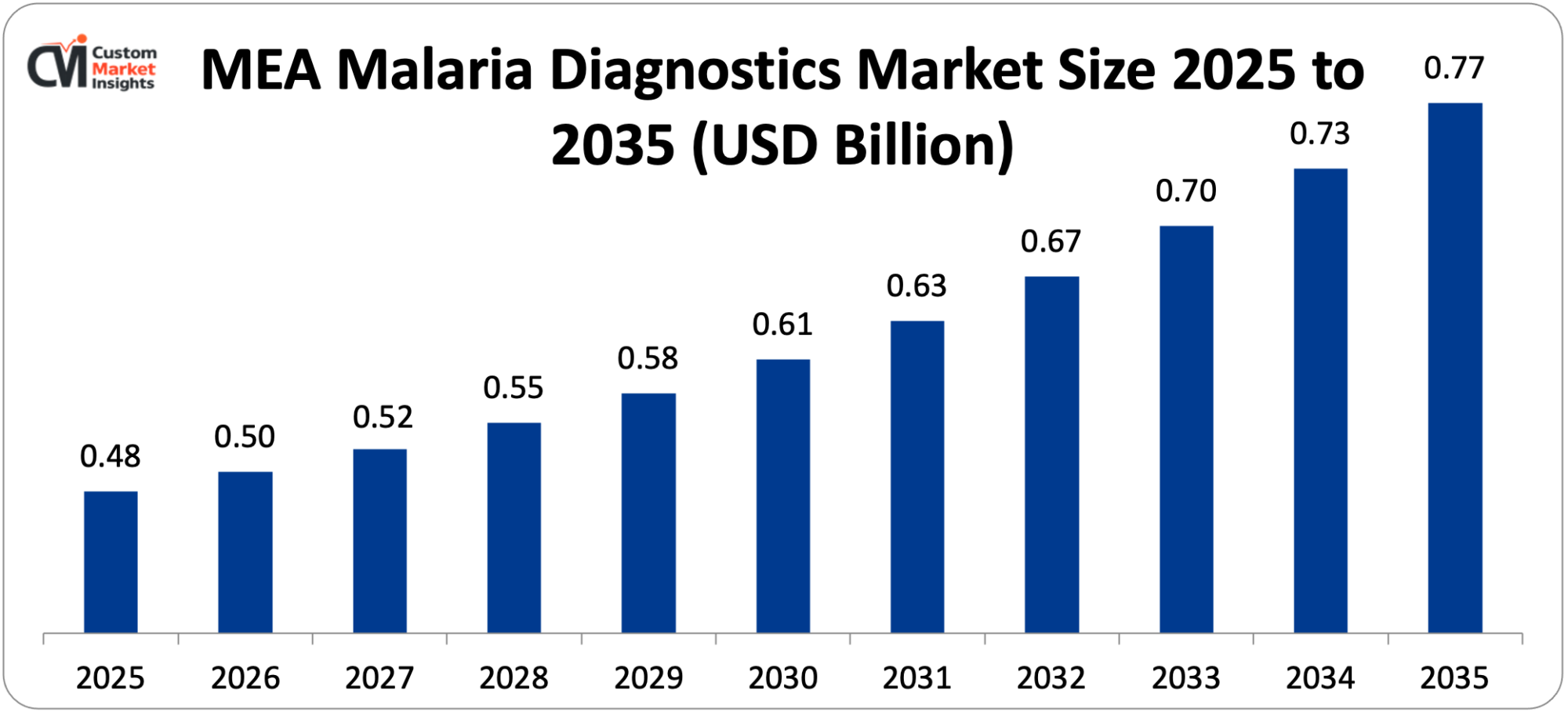
The Middle East & Africa malaria diagnostics market size is estimated at USD 477 million in 2025 and is projected to reach approximately USD 768 million by 2035, with a 5.1% CAGR from 2026 to 2035.
Why did Middle East & Africa Dominate the Market in 2025?
The most important player in the world is the Middle East & Africa, which is projected to have approximately 53% of the market share in 2025, owing to the fact that the region harbors over 90% of the world’s malaria burden, the African countries have the highest cases, there is wide distribution of Anopheles gambiae mosquitoes as the principal vectors, there is limited healthcare infrastructure that creates demand to access diagnostics, and finally, there is a high level of international aid to support malaria control programs. Preponderance of the disease is shown by the geographic concentration of the disease in the region, with Nigeria alone contributing 27% of the world cases, Democratic Republic of Congo (12%), Uganda (5%), and Mozambique (4%). The WHO and Global Fund focus their attention on African countries in the spending of the funds, including billions of dollars in diagnostic resources, purchasing drugs, and funding the programs.
Africa Market Trends
Africa in particular has been able to maintain significant growth through the development of new healthcare facilities and medical institutions in need of diagnostic devices, government interest in eradicating malaria with national strategic plans, international relations willing to offer technical and financial assistance, and community-based testing superimposing diagnostic access among remote residents. The heterogeneous epidemiology of the continent demands specific solutions, and the high-dissemination regions should have the large-volume diagnostic capacity, whereas the low-transmission areas should seek the elimination strategies with the consideration of sensitive diagnostics to detect asymptomatic carriers.
Why is Asia Pacific Experiencing Strong Growth?
The Asia-pacific region is performing well with a future estimated CAGR of 7.8% between 2026 and 2035 due to the high malaria burden in countries such as India, Indonesia, Myanmar and Papua New Guinea, a variety of species of parasites necessitating multi-species diagnostics, increased investment in health facilities by governments and increased use of advanced diagnostic technologies. The region has an estimated market value of USD 160.2 million in RDT amounting to about 36.4% of the global market. The sustained demand is caused by climate conditions that support the breeding of mosquitos and high populations of vulnerable classes. The National Vector Borne Disease Control Programme in India encourages the use of RDTs in rural regions, whereas Indonesia and Myanmar have strategies of elimination, which mandate increased diagnostics.
India Market Trends
The market presence of India is impressive due to the high number of people at risk (millions), the existence of various patterns of malaria transmission, the necessity to cover all malaria types with diagnostics, the highly developed governmental programs (free diagnosis and treatment), and the development of the sphere of healthcare by the private providers (advanced diagnostics). India is the heavy contributor of malaria in Asia as it makes strides towards eliminating malaria through better detection and treatment of the disease.
Why is North America Primarily Supporting Endemic Regions?
North America has few domestic malaria transmissions but is a very important source of diagnostic innovation, manufacturing, research funding and technical assistance to endemic areas. Major equipment and supplies used are produced by major diagnostic manufacturers such as Abbott, Beckman Coulter and Thermo Fisher Scientific who are found in the United States. The malaria control programs, such as the President Malaria Initiative launched by the U.S. government offers hundreds of millions of dollars in support of malaria control in countries where the disease is prevalent. Diagnostic research is undertaken in academic institutions to advance technology whereas regulatory bodies such as the FDA certify new diagnostics to meet quality standards.
Why is Europe Contributing Through Technology and Support?
North America has few domestic malaria transmissions but is a very important source of diagnostic innovation, manufacturing, research funding and technical assistance to endemic areas. Major equipment and supplies used are produced by major diagnostic manufacturers such as Abbott, Beckman Coulter and Thermo Fisher Scientific who are found in the United States. The malaria control programs, such as the President Malaria Initiative launched by the U.S. government offers hundreds of millions of dollars in support of malaria control in countries where the disease is prevalent. Diagnostic research is undertaken in academic institutions to advance technology whereas regulatory bodies such as the FDA certify new diagnostics to meet quality standards.
Top Players in the Market and Their Offerings
- Abbott Laboratories
- Access Bio Inc.
- Beckman Coulter Inc.
- Bio-Rad Laboratories Inc.
- Thermo Fisher Scientific Inc.
- Hoffmann-La Roche Ltd.
- Premier Medical Corporation Pvt. Ltd.
- Sysmex Corporation
- bioMérieux SA
- Siemens Healthineers
- Others
Key Developments
The market has undergone significant developments as participants seek to expand capabilities and enhance diagnostic access.
- In July 2022, Mylab Discovery Solutions launched the PathoDetect Extended Monsoon Fever Panel, a multiplex RT-PCR kit simultaneously detecting and differentiating malaria, dengue, chikungunya, Zika, Salmonellosis, Leptospirosis, and Leishmaniasis, expanding comprehensive fever diagnostics. (Source: Mylab Discovery Solutions)
- In February 2022, Kenya proposed locally produced PlamoCheck malaria kits and KEMCoV PCR COVID-19 kits by the Ministry of Health, where testing is cheaper and more accessible with production based locally. (Source: Kenya’s Ministry of Health)
These strategic activities have strengthened diagnostic accessibility, technological capabilities, and international cooperation, supporting global malaria elimination goals.
The Malaria Diagnostics Market is segmented as follows:
By Technology
- Rapid Diagnostic Tests (RDTs)
- Single Species Detection
- Multi-Species Detection
- Microscopy
- Light Microscopy
- Fluorescence Microscopy
- Molecular Diagnostic Tests
- Conventional PCR
- Real-Time PCR
- LAMP (Loop-Mediated Isothermal Amplification)
- Other Technologies
By Product Type
- Diagnostic Kits & Reagents
- Instruments
- Software & Services
By End-User
- Hospitals & Clinics
- Diagnostic Centers
- Academic & Research Institutes
- Others
Regional Coverage:
North America
- U.S.
- Canada
- Mexico
- Rest of North America
Europe
- Germany
- France
- U.K.
- Russia
- Italy
- Spain
- Netherlands
- Rest of Europe
Asia Pacific
- China
- Japan
- India
- New Zealand
- Australia
- South Korea
- Taiwan
- Rest of Asia Pacific
The Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of the Middle East & Africa
Latin America
- Brazil
- Argentina
- Rest of Latin America
Table of Contents
- Chapter 1. Report Introduction
- 1.1. Report Description
- 1.1.1. Purpose of the Report
- 1.1.2. USP & Key Offerings
- 1.2. Key Benefits For Stakeholders
- 1.3. Target Audience
- 1.4. Report Scope
- 1.1. Report Description
- Chapter 2. Market Overview
- 2.1. Report Scope (Segments And Key Players)
- 2.1.1. Malaria Diagnostics by Segments
- 2.1.2. Malaria Diagnostics by Region
- 2.2. Executive Summary
- 2.2.1. Market Size & Forecast
- 2.2.2. Malaria Diagnostics Market Attractiveness Analysis, By Technology
- 2.2.3. Malaria Diagnostics Market Attractiveness Analysis, By Product Type
- 2.2.4. Malaria Diagnostics Market Attractiveness Analysis, By End-User
- 2.1. Report Scope (Segments And Key Players)
- Chapter 3. Market Dynamics (DRO)
- 3.1. Market Drivers
- 3.1.1. High Disease Burden in Endemic Regions and Global Elimination Initiatives
- 3.1.2. Technological Advancements in Diagnostic Methods
- 3.2. Market Restraints
- 3.3. Market Opportunities
- 3.5. Pestle Analysis
- 3.6. Porter’s Forces Analysis
- 3.7. Technology Roadmap
- 3.8. Value Chain Analysis
- 3.9. Government Policy Impact Analysis
- 3.10. Pricing Analysis
- 3.1. Market Drivers
- Chapter 4. Malaria Diagnostics Market – By Technology
- 4.1. Technology Market Overview, By Technology Segment
- 4.1.1. Malaria Diagnostics Market Revenue Share, By Technology, 2025 & 2035
- 4.1.2. Rapid Diagnostic Tests (RDTs)
- 4.1.3. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.5. Key Market Trends, Growth Factors, & Opportunities
- 4.1.6. Single Species Detection
- 4.1.7. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.9. Key Market Trends, Growth Factors, & Opportunities
- 4.1.10. Multi-Species Detection
- 4.1.11. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.13. Key Market Trends, Growth Factors, & Opportunities
- 4.1.14. Microscopy
- 4.1.15. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.16. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.17. Key Market Trends, Growth Factors, & Opportunities
- 4.1.18. Light Microscopy
- 4.1.19. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.20. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.21. Key Market Trends, Growth Factors, & Opportunities
- 4.1.22. Fluorescence Microscopy
- 4.1.23. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.24. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.25. Key Market Trends, Growth Factors, & Opportunities
- 4.1.26. Molecular Diagnostic Tests
- 4.1.27. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.28. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.29. Key Market Trends, Growth Factors, & Opportunities
- 4.1.30. Conventional PCR
- 4.1.31. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.32. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.33. Key Market Trends, Growth Factors, & Opportunities
- 4.1.34. Real-Time PCR
- 4.1.35. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.36. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.37. Key Market Trends, Growth Factors, & Opportunities
- 4.1.38. LAMP (Loop-Mediated Isothermal Amplification)
- 4.1.39. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.40. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.41. Key Market Trends, Growth Factors, & Opportunities
- 4.1.42. Other Technologies
- 4.1.43. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 4.1.44. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.45. Key Market Trends, Growth Factors, & Opportunities
- 4.1. Technology Market Overview, By Technology Segment
- Chapter 5. Malaria Diagnostics Market – By Product Type
- 5.1. Product Type Market Overview, By Product Type Segment
- 5.1.1. Malaria Diagnostics Market Revenue Share, By Product Type, 2025 & 2035
- 5.1.2. Diagnostic Kits & Reagents
- 5.1.3. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 5.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.5. Key Market Trends, Growth Factors, & Opportunities
- 5.1.6. Instruments
- 5.1.7. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 5.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.9. Key Market Trends, Growth Factors, & Opportunities
- 5.1.10. Software & Services
- 5.1.11. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 5.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.13. Key Market Trends, Growth Factors, & Opportunities
- 5.1. Product Type Market Overview, By Product Type Segment
- Chapter 6. Malaria Diagnostics Market – By End-User
- 6.1. End-User Market Overview, By End-User Segment
- 6.1.1. Malaria Diagnostics Market Revenue Share, By End-User, 2025 & 2035
- 6.1.2. Hospitals & Clinics
- 6.1.3. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 6.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.5. Key Market Trends, Growth Factors, & Opportunities
- 6.1.6. Diagnostic Centers
- 6.1.7. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 6.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.9. Key Market Trends, Growth Factors, & Opportunities
- 6.1.10. Academic & Research Institutes
- 6.1.11. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 6.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.13. Key Market Trends, Growth Factors, & Opportunities
- 6.1.14. Others
- 6.1.15. Malaria Diagnostics Share Forecast, By Region (USD Billion)
- 6.1.16. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.17. Key Market Trends, Growth Factors, & Opportunities
- Malaria Diagnostics Market – Regional Analysis
- 6.2. Malaria Diagnostics Market Overview, By Region Segment
- 6.2.1. Global Malaria Diagnostics Market Revenue Share, By Region, 2025 & 2035
- 6.2.2. Global Malaria Diagnostics Market Revenue, By Region, 2025 – 2035 (USD Billion)
- 6.2.3. Global Malaria Diagnostics Market Revenue, By Technology, 2025 – 2035
- 6.2.4. Global Malaria Diagnostics Market Revenue, By Product Type, 2025 – 2035
- 6.2.5. Global Malaria Diagnostics Market Revenue, By End-User, 2025 – 2035
- 6.3. North America
- 6.3.1. North America Malaria Diagnostics Market Revenue, By Country, 2025 – 2035 (USD Billion)
- 6.3.2. North America Malaria Diagnostics Market Revenue, By Technology, 2025 – 2035
- 6.3.3. North America Malaria Diagnostics Market Revenue, By Product Type, 2025 – 2035
- 6.3.4. North America Malaria Diagnostics Market Revenue, By End-User, 2025 – 2035
- 6.3.5. U.S. Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.3.6. Canada Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.3.7. Mexico Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.3.8. Rest of North America Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.4. Europe
- 6.4.1. Europe Malaria Diagnostics Market Revenue, By Country, 2025 – 2035 (USD Billion)
- 6.4.2. Europe Malaria Diagnostics Market Revenue, By Technology, 2025 – 2035
- 6.4.3. Europe Malaria Diagnostics Market Revenue, By Product Type, 2025 – 2035
- 6.4.4. Europe Malaria Diagnostics Market Revenue, By End-User, 2025 – 2035
- 6.4.5. Germany Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.4.6. France Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.4.7. U.K. Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.4.8. Russia Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.4.9. Italy Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.4.10. Spain Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.4.11. Netherlands Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.4.12. Rest of Europe Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.5. Asia Pacific
- 6.5.1. Asia Pacific Malaria Diagnostics Market Revenue, By Country, 2025 – 2035 (USD Billion)
- 6.5.2. Asia Pacific Malaria Diagnostics Market Revenue, By Technology, 2025 – 2035
- 6.5.3. Asia Pacific Malaria Diagnostics Market Revenue, By Product Type, 2025 – 2035
- 6.5.4. Asia Pacific Malaria Diagnostics Market Revenue, By End-User, 2025 – 2035
- 6.5.5. China Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.5.6. Japan Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.5.7. India Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.5.8. New Zealand Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.5.9. Australia Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.5.10. South Korea Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.5.11. Taiwan Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.5.12. Rest of Asia Pacific Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.6. The Middle-East and Africa
- 6.6.1. The Middle-East and Africa Malaria Diagnostics Market Revenue, By Country, 2025 – 2035 (USD Billion)
- 6.6.2. The Middle-East and Africa Malaria Diagnostics Market Revenue, By Technology, 2025 – 2035
- 6.6.3. The Middle-East and Africa Malaria Diagnostics Market Revenue, By Product Type, 2025 – 2035
- 6.6.4. The Middle-East and Africa Malaria Diagnostics Market Revenue, By End-User, 2025 – 2035
- 6.6.5. Saudi Arabia Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.6.6. UAE Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.6.7. Egypt Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.6.8. Kuwait Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.6.9. South Africa Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.6.10. Rest of the Middle East & Africa Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.7. Latin America
- 6.7.1. Latin America Malaria Diagnostics Market Revenue, By Country, 2025 – 2035 (USD Billion)
- 6.7.2. Latin America Malaria Diagnostics Market Revenue, By Technology, 2025 – 2035
- 6.7.3. Latin America Malaria Diagnostics Market Revenue, By Product Type, 2025 – 2035
- 6.7.4. Latin America Malaria Diagnostics Market Revenue, By End-User, 2025 – 2035
- 6.7.5. Brazil Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.7.6. Argentina Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.7.7. Rest of Latin America Malaria Diagnostics Market Revenue, 2026 – 2035 (USD Billion)
- 6.1. End-User Market Overview, By End-User Segment
- Chapter 7. Competitive Landscape
- 7.1. Company Market Share Analysis – 2025
- 7.1.1. Global Malaria Diagnostics Market: Company Market Share, 2025
- 7.2. Global Malaria Diagnostics Market Company Market Share, 2024
- 7.1. Company Market Share Analysis – 2025
- Chapter 8. Company Profiles
- 8.1. Abbott Laboratories
- 8.1.1. Company Overview
- 8.1.2. Key Executives
- 8.1.3. Product Portfolio
- 8.1.4. Financial Overview
- 8.1.5. Operating Business Segments
- 8.1.6. Business Performance
- 8.1.7. Recent Developments
- 8.2. Access Bio Inc.
- 8.3. Beckman Coulter Inc.
- 8.4. Bio-Rad Laboratories Inc.
- 8.5. Thermo Fisher Scientific Inc.
- 8.6. F. Hoffmann-La Roche Ltd.
- 8.7. Premier Medical Corporation Pvt. Ltd.
- 8.8. Sysmex Corporation
- 8.9. bioMérieux SA
- 8.10. Siemens Healthineers
- 8.11. Others.
- 8.1. Abbott Laboratories
- Chapter 9. Research Methodology
- 9.1. Research Methodology
- 9.2. Secondary Research
- 9.3. Primary Research
- 9.3.1. Analyst Tools and Models
- 9.4. Research Limitations
- 9.5. Assumptions
- 9.6. Insights From Primary Respondents
- 9.7. Why Custom Market Insights
- Chapter 10. Standard Report Commercials & Add-Ons
- 10.1. Customization Options
- 10.2. Subscription Module For Market Research Reports
- 10.3. Client Testimonials
List Of Figures
Figures No 1 to 34
List Of Tables
Tables No 1 to 46
Prominent Player
- Abbott Laboratories
- Access Bio Inc.
- Beckman Coulter Inc.
- Bio-Rad Laboratories Inc.
- Thermo Fisher Scientific Inc.
- Hoffmann-La Roche Ltd.
- Premier Medical Corporation Pvt. Ltd.
- Sysmex Corporation
- bioMérieux SA
- Siemens Healthineers
- Others
FAQs
The key players in the market are Abbott Laboratories, Access Bio Inc., Beckman Coulter Inc., Bio-Rad Laboratories Inc., Thermo Fisher Scientific Inc., F. Hoffmann-La Roche Ltd., Premier Medical Corporation Pvt. Ltd., Sysmex Corporation, bioMérieux SA, Siemens Healthineers, Others.
The market is greatly influenced by government policies which include WHO prequalification to make sure that the quality of the diagnostic done meet the approved products that can be procured by international programs, national regulatory approvals to sell that specific diagnostic in the market which requires that a particular diagnostic be done to ensure that the antimalarial which is to be applied is taken up and that specifications are followed and also through quality assurance programs to detect the accuracy of the field diagnostic testing, treatment protocols that ensures that diagnostic is done before antimalarial is applied to prevent drug resistance and surveillance needs such as some countries may require mandatory Low-transmission countries require more sensitive diagnostics to identify the presence of an asymptomatic carrier, whereas high-transmission countries favor more accessible point-of-care testing to develop a variety of regulatory and programmatic demands that define product development and market potential.
Diagnostic pricing has a strong effect on access and the size of a program, with the typical cost of RDTs being less than USD 1 per unit to support large-scale use in resource-constrained environments, microscopy has initial equipment costs of USD 500-2,000 but low per-test costs and molecular diagnostics costs USD 10-50 per test, making it only cost-effective to use in reference labs and research environments. Some of the factors that enhance cost-effectiveness are volume purchasing by international organizations that realize economies of scale by obtaining a volume discount, local production by manufacturing in countries such as Kenya that reduce costs, donation programs where the U.S. government donates 1.7M free RDTs, and innovative financing sources such as Global Fund which funds the procurement of diagnostics. Economies of scale are obtained through high volume programs whereas higher prices on molecular tests are associated with high sensitivity that is worth the cost in elimination scenarios.
According to the current analysis, the market is expected to grow to be roughly USD 1.45 billion by 2035, with a steady growth owing to the high disease burden in endemic regions, the international commitment to eliminate malaria keeping funding and programs supportable, technology advancements in improving sensitivity and accessibility, continued expansion of molecular diagnostics application, integration of digital health providers to facilitate surveillance, and development of infrastructure to support healthcare in malaria-infested countries at 5.2% per annum between 2026 and 2035.
The Middle East & Africa will have the largest share, with approximately 53% of the total world market share, as the region has over 90% of the global malaria burden, six countries in Africa have 55% of the world malaria with Nigeria alone having 27%, and a large proportion of which is distributed widely with the Anopheles gambiae mosquitoes acting as the primary vectors, much international aid with WHO and Global Fund targeting African countries and the growing healthcare infrastructure, which needs diagnostic supplies and equipment to support large scale testing programs.
It is expected that the Asia-Pacific Region will record the highest CAGR of about 7.8% throughout the forecast period due to the high malaria burden in India, Indonesia and Myanmar, multi-species diagnostics as P. falciparum and P. vivax are circulating, government investment in health infrastructure; and the National Vector Borne Disease Control Programme in India which is encouraging the use of RDT as a part of elimination efforts and the region contributing 36.4% of the global RDT market.
It is also expected that the Global Malaria Diagnostics Market will grow because of the disease burden of 241 million cases and 627,000 deaths in 2020 with 95% in Africa, international efforts to eliminate malaria, such as WHO elimination 2025 which targets 25 countries; the large expenditure on the disease, with Nigeria receiving more than USD 2.6B in Global Fund and the UK contributing 206B shillings in Uganda; and technological innovations, such as ultra-sensitive RDTs with 5x.