Bacterial Nanocellulose Market Size, Trends and Insights By Form (Wet/Hydrogel Bacterial Nanocellulose (Native Pellicle, Hydrogel Sheet, Suspension), Dry Bacterial Nanocellulose (Freeze-Dried, Spray-Dried, Aerogel), Functionalized Bacterial Nanocellulose (Surface-Modified, Composite), Other Forms (Nanocrystalline, Nanofibrillated Suspension)), By Source (Komagataeibacter xylinus (formerly Acetobacter xylinum), Agrobacterium tumefaciens, Sarcina ventriculi, Other Sources (Rhizobium leguminosarum, Pseudomonas fluorescens, Engineered Strains)), By Application (Biomedical & Healthcare (Wound Dressings, Tissue Engineering, Drug Delivery, Diagnostics), Food & Beverage (Texturizer, Stabilizer, Fat Replacer, Dietary Fiber), Cosmetics & Personal Care (Skin Hydration, Mask, Active Delivery), Electronics & Sensors (Flexible Electronics, Biosensors, Energy Storage), Environmental Remediation (Heavy Metal Removal, Water Purification), Paper & Packaging (Barrier Coatings, Composite Reinforcement), Other Applications (Acoustic Materials, Filtration)), By End-Use Industry (Healthcare & Pharmaceuticals, Food & Beverage, Consumer Goods (Cosmetics, Personal Care), Electronics, Environmental & Industrial, Other Industries (Research, Defense, Textiles)), and By Region - Global Industry Overview, Statistical Data, Competitive Analysis, Share, Outlook, and Forecast 2026 – 2035
Report Snapshot
| Study Period: | 2026-2035 |
| Fastest Growing Market: | Asia Pacific |
| Largest Market: | North America |
Major Players
- Cellutech AB
- Nanovia SAS
- BioMedical Systems Inc.
- Nanotech Finland Oy
- Others
Reports Description
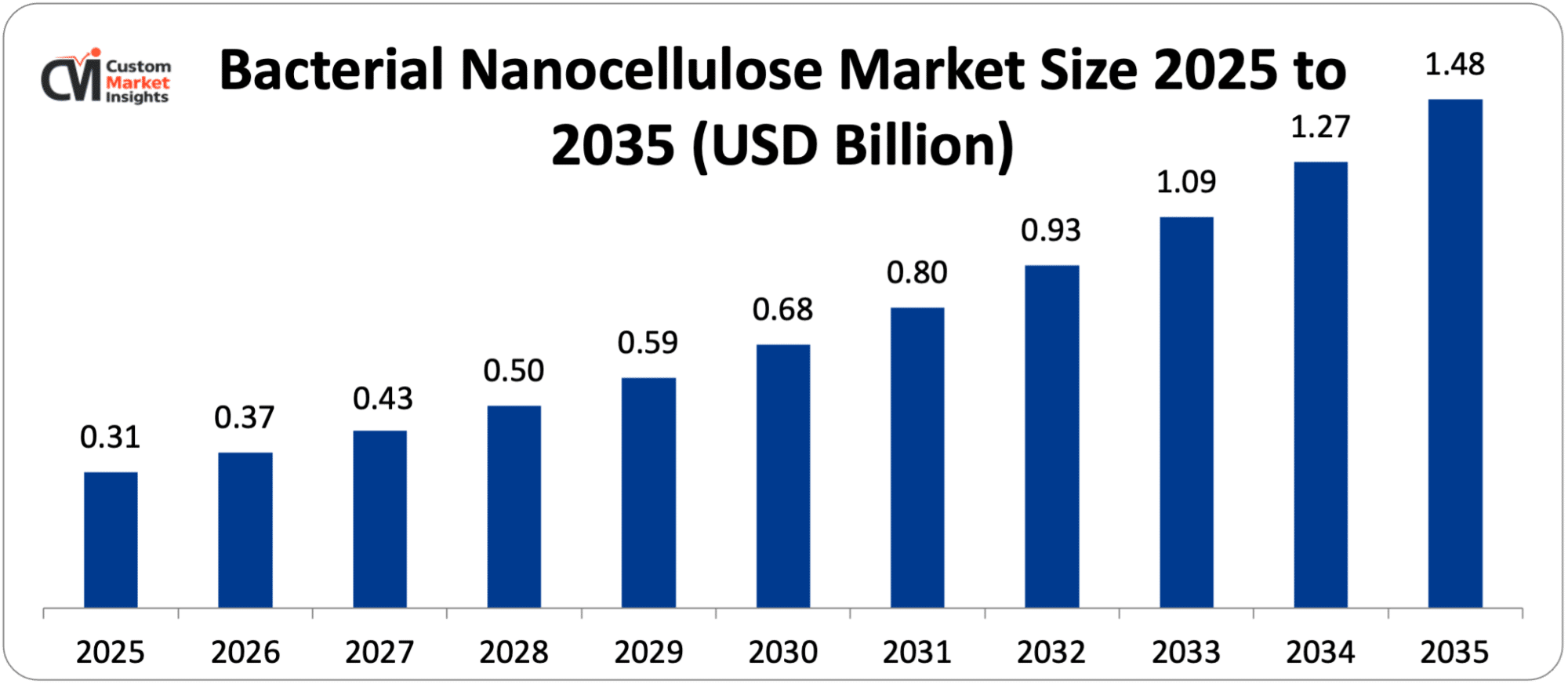
The world market scale of bacterial nanocellulose is estimated at USD 0.31 billion in 2025 and will grow by USD 0.37 billion in 2026 and then grow by around USD 1.48 billion by 2035 with a CAGR of 14.9% in 2026 to 2035.
The swiftly growing commercial use of bacterial nanocellulose in wound care and biomedical scaffold applications where BNC has demonstrated its extraordinary capacity to combine high water-holding capacity, mechanical strength, biocompatibility, and three-dimensional nanofibrillar network structure to confer clinical performance advantages over more conventional wound dressing and tissue engineering products, shows that no human synthetic or plant-derived materials can match its performance. BNC is progressively finding applications in the food industry as a clean label texturizing, stabilizing, and fat-replacing ingredient, the growing trend in the cosmetics and personal care industry adoption of BNC as a high-performance skin hydration and active ingredient delivery matrix in premium skincare formulations, the progressive scale-up of BNC fermentation production technology reducing manufacturing cost toward commercial competitiveness with alternative specialty biomaterials, and the emerging applications of BNC in flexible electronics, biosensors, and composite materials driven by BNC’s exceptional nanomechanical properties and surface chemistry versatility collectively drive robust and sustained market growth throughout the forecast period.
Market Highlight
- North America is the leader in the bacterial nanocellulose market with 36% market share in 2025.
- Asia Pacific will grow at a high CAGR of 17.8% in the years 2026 to 2035.
- Formatically, the wet/hydrogel bacterial nanocellulose section had taken about 58% of the market share in 2025.
- By form, the dry bacterial nanocellulose segment is increasing at a greater CAGR of 16.4% between 2026 and 2035.
- By source, the Komagataeibacter xylinus segment will have the largest share of 68% in the market in 2025, but other sources will grow at the highest CAGR of 18.2% within the estimated time between 2026 and 2035.
- By application, the biomedical and healthcare segment will have the largest market share of 42% in 2025, and the electronics and sensors segment will have the highest rate of CAGR of 21.6% between 2026 and 2035.
- By end-use industry, the healthcare and pharmaceuticals segment will have the largest share of 44% in 2025, and the electronics segment will have the greatest CAGR of 22.4% between 2026 and 2035.
Significant Growth Factors
The Bacterial Nanocellulose Market Trends present significant growth opportunities due to several factors:
- Biomedical Application Expansion Driving Premium Market Value and Clinical Adoption:
The clinical validation and commercial uptake of bacterial nanocellulose in a growing variety of biomedical applications, such as chronic wound care, burn wound care, periodontal guided tissue regeneration, eardrum reconstruction, vascular graft development, and as a drug delivery matrix, is engineering BNC as a commercially viable biomedical material platform whose clinical performance benefits over conventional materials translate into premium product positioning, high reimbursement opportunities, and long term commercial growth through reported patient outcome improvements.
The wound care application is the most commercially developed and most revenue-generating biomedical division of BNC wound dressings, which are shown to be superior in all the clinical parameters of moisture retention, mechanical protection, conformability, bacterial barrier functionality, and accelerated healing required, which all combine to form the clinical outcomes and overall treatment cost of wound management. There is extensive published clinical evidence data — in chronic venous leg ulcers, pressure ulcers, diabetic foot ulcers, and partial-thickness burns, all of which impact tens of millions of patients per year in developed healthcare systems — of a better healing rate, less pain with each dressing change, fewer dressing changes per healing event, and more patient-reported comfort scores with BNC dressings than with an alternative standard-of-care dressing.
The worldwide chronic wound problem, including published epidemiology estimates of 2.5% of all U.S. population having a chronic wound at any one time, equal to an estimated 8.2 million Americans, and the enormous economic costs of treating chronic wounds, over USD 25 billion in annual U.S. healthcare spending, provide an addressable market of large scale to justify a sizable investment in BNC wound care product development by medical device and wound care companies. The use of BNC in tissue engineering scaffolds, where the nanofibrillar network architecture of BNC scaffolds is patterned to resemble the native extracellular environment of the tissue that the cells should regenerate, where the BNC scaffolds use of high water content has preserved the hydrated microenvironment on which cellular viability depends, and where functionality of BNC scaffolds through surface modification has made it versatile in changes of growth factor, adhesion peptide and bioactive molecule presentation to cells and tissue regeneration, is moving out of research proof-of The ear drum reconstruction application of BNC, which was invented and licensed by BioMedical Systems and Ear Technology Corporation, has undergone regulatory clearance and clinical implementation in tympanoplasty procedures in which BNC membrane is used as biocompatible scaffold of tympanic membrane regeneration with superior clinical results when compared with traditional autologous tissue grafts in published comparative clinical studies that show a successful niche commercial BNC biomedical product which demonstrates the regulatory pathway and clinical validation process that applies to more broad BNC biomedical applications.
- Clean Label Food Ingredient Demand and Functional Food Applications:
The increasing popularity of clean label food products in the world market, where food producers are reformulating their products to replace synthetic emulsifiers, stabilizers, thickeners, and fat replacers with naturally derived ingredient alternatives that consumers believe to be healthier, more recognizable, and more trustworthy, is providing commercial opportunity to bacterial nanocellulose as a high value natural food ingredient whose bio-based origin, cellulose backbone structure, and superior functional performance place it in the clean label ingredient space that is actively under construction by food manufacturers. FNC functional properties in food, such as extraordinary water-holding properties allowing moisture retention in baked foods and meat items without the use of synthetic humectants, emulsion stabilization through interfacial adsorption at oil-water interfaces equivalent to synthetic emulsifiers at lower concentrations, the formation of gel to provide texture and mouthfeel enhancement in dairy alternatives and low-fat foods, and suspension stabilization to maintain the dispersion of particles in beverages and liquid food products. — jointly meet the formulation issues that food manufacturers encounter in reformulating clean label products using synthetic functional ingredients that are commonly regarded as conventional.
The alternative protein food and plant-based sector, one of the most rapidly growing and highly innovation-driven sectors of the food industry, provides an especially attractive BNC food application opportunity, since the fibrous texture, control of moisture, and fat replacement properties of BNC allow it to resolve the main formulation difficulties of developing plant-based meat, dairy, and seafood alternative products with organoleptic characteristics equal to those of their animal-based counterparts. The fat replacement application where BNC hydrogel at 0.5-2.0% incorporated into food formulations can substitute 20-40% of the fat content and still provide the same texture, mouthfeel, and sensorial attributes in reduced-fat dairy products, dressings, and spreads, which forms a clean label fat replacement ingredient positioning that commands high prices in comparison to conventional fat replacers such as maltodextrin and modified starch, which do not have the clean label credentials and the same functional performance of BNC.
The scientific basis of regulatory submission and commercial product development that major food companies are pursuing based on research investigation and moving towards product development through research introduction bottom and above current approved food ingredient usage levels is published research on the experiments of the Technical University of Denmark and Nestle. Research on the functional performance of BNC in yogurt, mayonnaise, and plant-based meat products reports a performance with regard to texture and stability measurable in equivalent or greater values than conventional food ingredients at the concentration levels when compared and contrasted with established approved limits of food ingredient use.
What are the Major Advances Changing the Bacterial Nanocellulose Market Today?
- Fermentation Process Engineering and Production Cost Reduction Enabling Commercial Scale: The long-term improvement of the bacteria nanocellulose fermentation process engineering including the optimization of bioreactor design to produce BNC; the optimization of carbon source and nitrogen source to lower the cost of the medium, fed-batch and the continuous fermentation strategy to enhance the volumetric productivity and optimization of oxygen transfer to overcome the dissolved oxygen limitation that limits the BNC pellicle thickness and the production rate in the static culture, and finally, the downstream processing streamlining to reduce the cost of burden of purification, is steadily narrowing the production cost gap between BNC and other special That basic issue of scale-up of BNC fermentation is based on the pellicle-based growth behavior of Komagataeibacter xylinus, which forms BNC at the air-liquid interface in static culture, which has historically made BNC production limited to low-voltage fermentation in shallow tray fermentation systems with low volumetric productivity and requiring much labor to operate tray fermentation systems, thus limiting large scale manufacturing and limiting ingredient costs to only high-value medical and cosmetic uses. The development of agitated bioreactor (BNC) production – where bioreactor design increases such as rotating disk systems, airlift systems, or specially designed impeller shapes, ensuring adequate dissolved oxygen transfer with little shear of the hydrodynamic system disrupting cellulose nanofiber network formation – has enabled the BNC production in common stir tank bioreactors with volumetric productivities of 0.1-0.5 g/L/hour that are 5-10 fold better than the traditional stir tank bioreactor, greatly lowering the capital and operating cost per The manufacturability of alternative low-cost carbon sources to BNC fermentation – the costly glucose medium employed in laboratory-scale production has been replaced with agricultural and industrial waste streams as sources of inexpensive carbon – is making possible the economics of manufacturability of BNC fermentation needed in manufacturing food and commodity applications, where ingredient cost sensitivity is significantly greater than in high-end medical applications. The viability of waste-stream-based BNC production with low costs as illustrated by published reports on fermentation optimization studies by academic research groups, such as the work of Universidade de São Paulo on BNC production using sugarcane molasses and the University of Aveiro on BNC production using olive mill wastewater, shows that the industry is increasingly moving in the direction of commercial-scale implementation of waste-stream-based BNC production. The large-scale BNC food ingredient production in China of Hainan Yida Food Industrial Group, one of the most commercially developed food-grade BNC manufacturing facilities in the world, with a production capacity estimated to be hundreds of tonnes/annum, portrays the fact that production of low-cost BNC food ingredients is technically viable at large scale.
- BNC Surface Functionalization and Composite Material Development: The versatility BNC can now be applied to, in addition to its material properties, has expanded dramatically because of the development of chemical and biological methods of surface functionalizing bacterial nanocellulose – allowing modification of the surface hydroxyl groups of BNC to introduce a wide range of functional chemistries such as amine, carboxyl, aldehyde, phosphate, and attaching biomolecule functionalities – which are creating functionalized BNC analogs with custom bioactivity, electronic conductivity, optical characteristics, and chemical selectivity which BNC alone cannot provide when required in The surface of BNC is functionalized with hydroxyl groups that can be chemically modified through well-established cellulose chemistry reactions such as TEMPO-mediated cellulose oxidation to add carboxyl groups, periodate oxidation to add aldehyde groups to allow Schiff base conjugation of amine-containing molecules, silylation to add hydrophobic silyl groups and esterification to add hydrophobic aliphatic groups – collectively offering a wide range of surface chemistry reactions to bring specific functional characteristics to BNC whilst maintaining One of the most commercially critical unmet needs in the wound care market, which is currently being approached by the antimicrobial BNC development, whereby silver nanoparticles, copper nanoparticles, zinc oxide nanoparticles or antimicrobial peptides are immobilized on BNC nanofiber surfaces via in situ nanoparticle development within the BNC hydrogel or via surface conjugation chemistry, is the antimicrobial wound dressings of infected chronic wounds; which is a large and high-end market niche that normal BNC wound dress The antimicrobial performance attestation of antimicrobial BNC wound care products is being supplied by published findings of antimicrobial BNC wound dressing development programs with regulated release of antimicrobial agents over 714 days of BNC maturity with bactericidal properties against the methicillin-resistant Staphylococcus aureus plus additional chronic wound pathogens that a number of firms are in the process of submitting to regulatory approval. Conductive BNC Composites Conductive BNC composites are electronic materials in which the electroactive materials (carbon nanotubes, graphene oxide, polyaniline, PEDOT:PSS conducting polymers, etc.) are incorporated into BNC scaffolds, and these electronic materials have enabled flexible electronic applications such as wearable biosensors, electronic skin, biodegradable transient electronics, and piezoelectric energy harvesting devices that make use of the mechanical flexibility, high surface area, and biodegradability of BNC.
- In Situ BNC Production and 3D Bioprinting Integration: New groups of BNC-based biomedical materials whose behavior cannot be obtained through standard ex situ methods of BNC production followed by insertion into the final product form are being developed through the development of in situ bacterial nanocellulose production strategies – in which Komagataeibacter bacteria are directly incorporated into hydrogel systems or in 3D bioprinting inks or cell culture scaffolds where they produce BNC nanofibers in situ during incubation to provide living composite materials with constantly evolving nanofiber network architecture. In situ production of BNCs in hydrogel matrices – as shown in research programs utilizing alginate, gelatin and polyethylene glycol hydrogel hosts – produces interpenetrating network-based composites with BNC nanofibers synthesized in situ strengthening the surrounding gel matrix and radically changing mechanical properties without the need to process pre-formed BNC which tends to disrupt the nanofiber network architecture. The 3D bioprinting integration – where Komagataeibacter bacteria are encapsulated in biocompatible ink formulations compatible with extrusion based bioprinting setups, allowing complex three dimensional structures to be printed which are then incubated to form the BNC nanofibers within the printed structure – somehow couples the design freedom of 3D bioprinting with the biological performance properties of BNC to allow the printing of anatomically shaped bio wound dressings, patient specific tissue engineering scaffolds, and complex microfluidic device substrates with BNC The Living Materials concept, where bacteria-containing BNC composites retain bacterial viability and BNC production functionality over prolonged periods allowing continuous self-recovery of mechanical properties and adopting responses to environmental stimuli, is the research frontier of BNC material science, with proofs of concept in the literature showing self-healing mechanical properties and dynamic responses to environmental stimuli and allowing entirely new application categories of BNC materials in soft robotics, environmental monitoring and autonomous biomedical devices. Studies published by Empa (Swiss Federal Laboratories of Materials Science and Technology) and MIT have shown the possibility of 3D-printing bacteria-containing BNC composites with controlled architectural complexity, which provides the scientific basis for the manufacture of scale-up programs to realize the early commercial applications of these living material concepts in wound dressing customisation and tissue engineering scaffold fabrication.
Category Wise Insights
By Form
Why Does Wet/Hydrogel Bacterial Nanocellulose Lead the Market?
Wet and hydrogel bacterial nanocellulose represents the highest unit of the total market share at about 58% in 2025 and this is the direct commercial use of BNC in its native biosynthetic format: as-produced hydrogel pellicles, membranes, and molded hydrogel pieces containing 97 -99% water with the undisturbed three-dimensional nanofiber network structure that gives BNC its characteristic properties – in the wound care, cosmetic mask, food ingredient and biomedical scaffold applications that represent the biggest In wet BNC form, the original structure of the nanofiber network in biosynthesis has not been destroyed by the mechanical and structural perturbations of the drying process and is thus intact, retaining all the expression of BNC of being a water-holding structure, mechanically compliant, and biologically interactive. Bio Fill Produtos Biotecnologicos in Brazil, BIONEXT of the Brazilian biomedical industry, and other developers of the European and North American markets promote commercial BNC wound dressing in the wet hydrogel form, which provides wound contact layers with the moisture-balancing and mechanical cushioning effect that the intact BNC hydrogel structure brings. The cosmetic BNC mask application, where wet BNC pellicle sheets are used as the mask of hydrating and active ingredient delivery to the facial skin, is the wet BNC application with the highest unit count volume (where skin mask applications constitute the highest volume of wet BNC applications), and the Asian cosmetic mask market is a major consumer of the BNC in terms of unit quantities when used as a skin mask with the attractive consumer story of nanoscale bacterially biosynthesized cellulose nanofibers.
By Source
Why Does Komagataeibacter xylinus Lead the Market?
Komagataeibacter xylinus (previously Acetobacter xylinum and Gluconacetobacter xylinus) is the dominant BNC production system, with a market share of about 68 in 2025, based on decades of scientific research and optimization of processes, strain engineering and commercial production history with this organism that has put it on the standard reference list of fermentation biology, best formulations of production media, and best known downstream processing protocols. K. xylinus is the highest-specific-productivity producer of BNC and best-uniform-morphology nanofiber among natural BNC-producing organisms, the individual cellulose nanofiber diameter of 20100 nm and 7090% crystallinity index are two structural properties that define the best material performance of BNC in all application sectors; the result is that BNC produced by K. xylinus has become the reference material whose characteristics are the basis of BNC’s commercial value proposition in all its application markets. The proximity of commercial BNC production infrastructure to K. xylinus fermentation products implies that experience of scale-up in production, both in data on equipment validation and regulatory documentation, as well as in supply chain relationships, are all structured around this organism, which establishes a strong practical advantage to commercial BNC production that other organisms will need to leap to produce similarly on a commercial scale.
By Application
Why Does Biomedical & Healthcare Lead the Market?
The area with the highest application market is biomedical and healthcare at about 42% of the total market share in 2025 due to the convergence of the superior performance credentials of BNC in the wound care market, tissue engineering, and drug delivery products due to their biological performance, the premium price that can be charged on medicine driven wound care products, and the existing body of clinical evidence base accrued over decades of research and commercial use of BNC wound care products in Brazil, Europe, and Asia that provides the documentation background to regulatory approval of new BNC medical device products. The wound care application covers the entire chronic wound management range, including acute burn wound management, venous leg ulcer management, and diabetic foot ulcer management, as well as pressure ulcer management with benefits of BNC dressings showing across-the-board benefits in all wound types due to its unique combination of moisture retention, bacterial barrier activity, ability to conform to irregular wound geometries, and non-adherence to the wound bed that contributes to trauma during dressing change. The most rapidly growing is the electronics and sensors segment with a CAGR of 21.6% between 2026 and 2035, as the flexible and wearable electronics market demands biodegradable, biocompatible, and mechanically flexible substrate materials in the next generation of body-worn sensors, implantable transient electronics, and environmentally friendly electronic items that demand various unique BNC attributes of nanomechanical strength, optical transparency, versatility of surface functionalization, and biodegradability that conventional electronic substrate materials such as PET film and PI film cannot satisfy.
By End-Use Industry
Why Does Healthcare & Pharmaceuticals Lead the Market?
The largest end-use market at about 44% of market share in 2025 is healthcare and pharmaceuticals, indicating that the highest value BNC commercial applications are concentrated in the medical devices and pharmaceutical products sectors where clinical performance benefits receive the highest possible prices of USD 50 to USD 500 per unit in wound care applications, USD 200-USD 2000 per implant in tissue engineering scaffold applications and USD 500-USD 5000 per kilogram in pharmaceutical-grade BNC excipient. The electronics segment is the fastest growing end-use industry with an outlook of 22.4% CAGR between 2026 and 2035, as it reflects the new but rapidly expanding commercial uses of BNC in flexible electronics where a small number of early commercial products that are using BNC substrates and composites are already commercially viable, and a significantly larger pipeline of research and development programs are in progress to commercialisation across wearable health sensors, biodegradable Internet of Things systems and sustainable display substrate applications.
Report Scope
| Feature of the Report | Details |
| Market Size in 2026 | USD 0.37 billion |
| Projected Market Size in 2035 | USD 1.48 billion |
| Market Size in 2025 | USD 0.31 billion |
| CAGR Growth Rate | 14.9% CAGR |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Key Segment | By Form, Source, Application, End-Use Industry and Region |
| Report Coverage | Revenue Estimation and Forecast, Company Profile, Competitive Landscape, Growth Factors and Recent Trends |
| Regional Scope | North America, Europe, Asia Pacific, Middle East & Africa, and South & Central America |
| Buying Options | Request tailored purchasing options to fulfil your requirements for research. |
Regional Analysis
How Big is the North America Market Size?
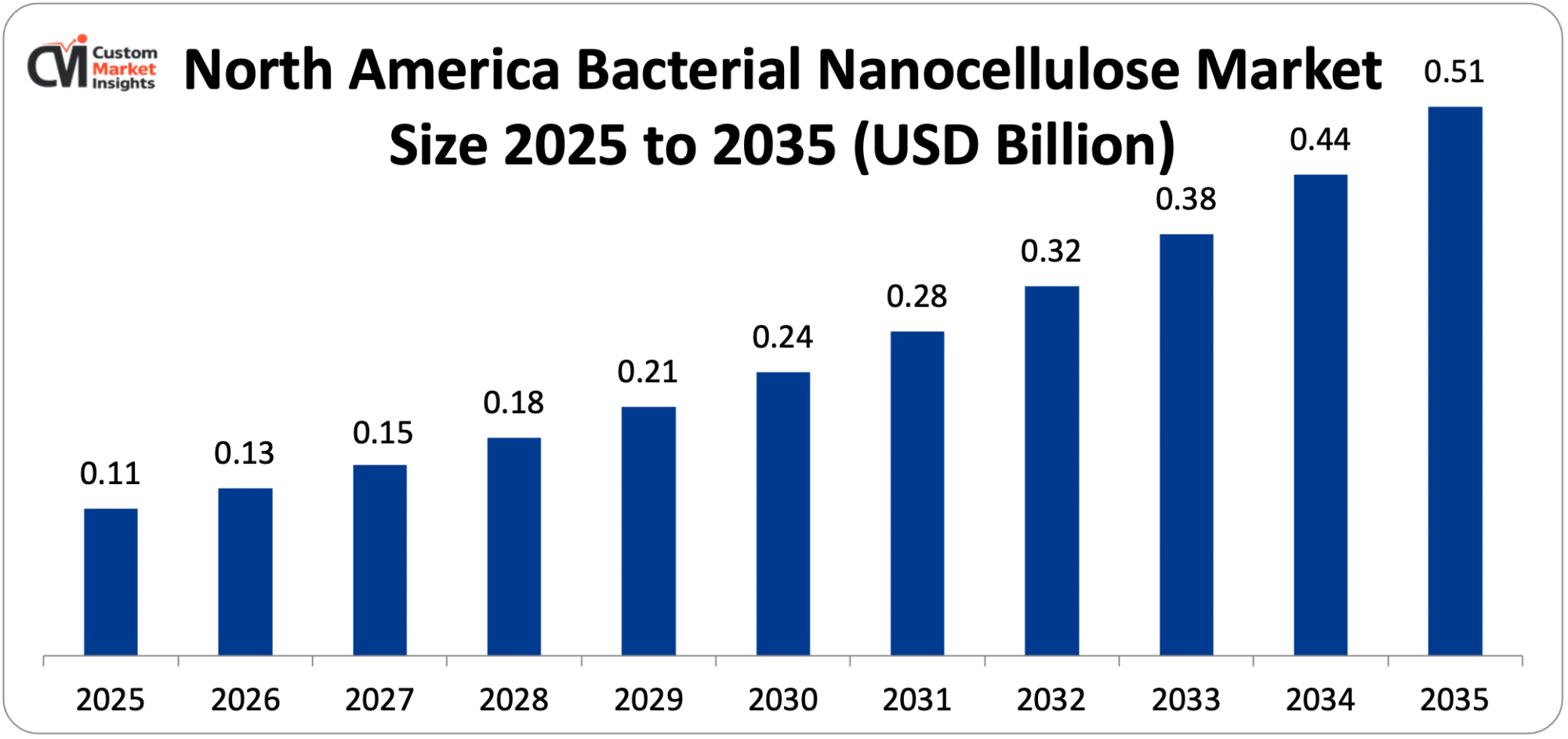
The North America bacterial nanocellulose market size is estimated at USD 112 million in 2025 and is projected to reach approximately USD 512 million by 2035, with a CAGR of 16.4% from 2026 to 2035.
Why Did North America Dominate the Market in 2025?
In 2025, the market share of the BNC biomedical innovation in the United States is about 36% of the market, indicating the United States position as the largest advanced wound care market in the world, with the U.S. chronic wound management product market estimated at over USD 25 billion annually, representing the largest addressable market opportunity of premium BNC wound care products. the concentration of the world’s leading biomaterials and medical device research and development programs at universities and research institutes in United States and the FDA regulatory framework is offering the best established channel of approval of BNC medical device products. The University of British Columbia, University of Toronto, and National Research Council Canada, Canadian research institutes, have come up with key BNC research initiatives in the biomedical and functional materials sectors, with Axcelon Biopolymers Corporation being the most commercially developed BNC developer in the biomedical arena. The active interest of the U.S. food industry in BNC as a clean label ingredient, as evidenced by the BNC ingredient assessment programs implemented by major food companies (such as Nestle, Unilever, and General Mills) as part of their clean label reformulation efforts, is establishing a growing food ingredient BNC demand that will form a large market niche as BNC regulatory approval pathways through FDA GRAS notification become available as food grade BNC.
Why is Europe a Strategically Important Market?
It has been estimated that the European bacterial nanocellulose market will be about USD 78 million by 2025 and is projected to be about USD 346 million by 2035 with a CAGR of 16.1%. The European market is a market of fundamental strategic significance because it is also the location of a number of the leading BNC research and commercialization programs – with Cellutech AB in Sweden, Nanotech Finland in Finland, and Nanovia in France being established European BNC companies and major academic BNC research programs at institutions such as the University of Helsinki, KTH Royal Institute of Technology, and Empa Switzerland; the regulatory framework of the European Union on advanced wound dressing and biomaterials supplying the CE marking route to commercially approve the BNC medical device, and the The largest national European BNC market opportunity, the advanced wound care market in Germany which is one of the largest markets in Europe, as well as the strong research culture of biomaterials in Germany with universities such as RWTH Aachen and the Fraunhofer institutes. The policy priorities of the EU bioeconomy of green materials and circular bioeconomy, including the EU Bioeconomy Strategy and the support of bio-based material development provided in the frames of the Horizon Europe research programs, provide the favorable policy climate for BNC that is based on biotechnology and is a renewable material that meets the European industrial policy goals.
Why is Asia Pacific the Fastest-Growing Market?
The Asian Pacific has the most active regional market with an expected CAGR of 17.8% between 2026 and 2035 due to the established commercial BNC food ingredient manufacturer in China, which is the leading source of commercial BNC products in the Asia Pacific, with Hainan Yida Food Industrial Group proving the commercial-scale production capability that is prompting the development of more advanced BNC production capacity of the Chinese market and the active development of commercial BNC wound dressing in the markets of China, Japan, and South Korea, Japan having a developed biomaterials and cos. The BNC Lab and BNC Research Institute of South Korea are focused on BNC research and commercialization activities whose products include academic publications on BNC science as well as commercial product development programs in the areas of wound care, cosmetics and food applications in the Korean local and export markets. The cosmetic ingredient market in Japan, which is one of the most technically advanced and scientifically substantiated in the world, is considering the use of BNC in several skin care application formats as Shiseido, Kose, and Pola Orbis, leading Japanese cosmetic companies, are carrying out BNC ingredient performance assessment programs.
Why is Brazil an Emerging Market with Unique Strategic Importance?
Brazil has a niche in the international BNC market in that it is the home nation of the longest history of commercial BNC wound care deployment in the world, with Biofill R BNC wound dressings and Bio Fill Produtos Biotecnologicos commercializing BNC wound care products in Brazil in the 1980s, becoming the first commercial BNC wound care product worldwide, putting Brazil in the position of being a reference market in BNC wound care clinical evidence and as a manufacturing location with an established history of production in BNC wound care products. The tropical climate of Brazil offers the best fermentation environment for producing K. xylinus using local and cheap sources of agricultural carbon such as sugarcane molasses and fruit processing waste, which makes the economics of production of BNC competitive with other and more costly production locations in the north. The research efforts of Brazilian scholars at the University of São Paulo, University of Campinas (UNICAMP), and Embrapa have produced a massive published body of BNC fermentation optimization, development of applications, and scale-up that gives reference knowledge to the world on developing BNC production processes.
Top Players in the Market and Their Offerings
- Axcelon Biopolymers Corporation
- Hainan Yida Food Industrial Group Co. Ltd.
- Cellutech AB
- Nanovia SAS
- BioMedical Systems Inc.
- Nanotech Finland Oy
- Bowil Biotech
- Nanologica AB
- Creative Medical Technology Holdings Inc.
- BNC Lab (South Korea)
- Biofabrication Lab (Brazil)
- Others
Key Developments
The market has undergone significant developments as industry participants seek to advance BNC production scale-up, expand biomedical product regulatory approvals, and respond to the growing commercial interest from the food, cosmetics, and electronics industries globally.
- In February 2026: Pandorum Technologies announced the successful completion of a CAD 18 million Series B financing round -led by Protons Corporate, with participation by BDC Capital and strategic investors in the advanced wound care and biomaterials industry—to scale up its BNC production facility in Vancouver, British Columbia.
- In January 2025: Cellutech AB declared a business licensing settlement with a large European cosmetic ingredient purchaser – granting them the distribution legalization of CelluForce BNC nanocellulose suspension in 18 European markets in cosmetic applications.
The Bacterial Nanocellulose Market is segmented as follows:
By Form
- Wet/Hydrogel Bacterial Nanocellulose (Native Pellicle, Hydrogel Sheet, Suspension)
- Dry Bacterial Nanocellulose (Freeze-Dried, Spray-Dried, Aerogel)
- Functionalized Bacterial Nanocellulose (Surface-Modified, Composite)
- Other Forms (Nanocrystalline, Nanofibrillated Suspension)
By Source
- Komagataeibacter xylinus (formerly Acetobacter xylinum)
- Agrobacterium tumefaciens
- Sarcina ventriculi
- Other Sources (Rhizobium leguminosarum, Pseudomonas fluorescens, Engineered Strains)
By Application
- Biomedical & Healthcare (Wound Dressings, Tissue Engineering, Drug Delivery, Diagnostics)
- Food & Beverage (Texturizer, Stabilizer, Fat Replacer, Dietary Fiber)
- Cosmetics & Personal Care (Skin Hydration, Mask, Active Delivery)
- Electronics & Sensors (Flexible Electronics, Biosensors, Energy Storage)
- Environmental Remediation (Heavy Metal Removal, Water Purification)
- Paper & Packaging (Barrier Coatings, Composite Reinforcement)
- Other Applications (Acoustic Materials, Filtration)
By End-Use Industry
- Healthcare & Pharmaceuticals
- Food & Beverage
- Consumer Goods (Cosmetics, Personal Care)
- Electronics
- Environmental & Industrial
- Other Industries (Research, Defense, Textiles)
Regional Coverage:
North America
- U.S.
- Canada
- Mexico
- Rest of North America
Europe
- Germany
- France
- U.K.
- Russia
- Italy
- Spain
- Netherlands
- Rest of Europe
Asia Pacific
- China
- Japan
- India
- New Zealand
- Australia
- South Korea
- Taiwan
- Rest of Asia Pacific
The Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of the Middle East & Africa
Latin America
- Brazil
- Argentina
- Rest of Latin America
Table of Contents
- Chapter 1. Report Introduction
- 1.1. Report Description
- 1.1.1. Purpose of the Report
- 1.1.2. USP & Key Offerings
- 1.2. Key Benefits For Stakeholders
- 1.3. Target Audience
- 1.4. Report Scope
- 1.1. Report Description
- Chapter 2. Market Overview
- 2.1. Report Scope (Segments And Key Players)
- 2.1.1. Bacterial Nanocellulose by Segments
- 2.1.2. Bacterial Nanocellulose by Region
- 2.2. Executive Summary
- 2.2.1. Market Size & Forecast
- 2.2.2. Bacterial Nanocellulose Market Attractiveness Analysis, By Form
- 2.2.3. Bacterial Nanocellulose Market Attractiveness Analysis, By Source
- 2.2.4. Bacterial Nanocellulose Market Attractiveness Analysis, By Application
- 2.2.5. Bacterial Nanocellulose Market Attractiveness Analysis, By End-Use Industry
- 2.1. Report Scope (Segments And Key Players)
- Chapter 3. Market Dynamics (DRO)
- 3.1. Market Drivers
- 3.1.1. Biomedical Application Expansion Driving Premium Market Value and Clinical Adoption
- 3.1.2. Clean Label Food Ingredient Demand and Functional Food Applications
- 3.2. Market Restraints
- 3.3. Market Opportunities
- 3.5. Pestle Analysis
- 3.6. Porter Forces Analysis
- 3.7. Technology Roadmap
- 3.8. Value Chain Analysis
- 3.9. Government Policy Impact Analysis
- 3.10. Pricing Analysis
- 3.1. Market Drivers
- Chapter 4. Bacterial Nanocellulose Market – By Form
- 4.1. Form Market Overview, By Form Segment
- 4.1.1. Bacterial Nanocellulose Market Revenue Share, By Form, 2025 & 2035
- 4.1.2. Wet/Hydrogel Bacterial Nanocellulose (Native Pellicle, Hydrogel Sheet, Suspension)
- 4.1.3. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 4.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.5. Key Market Trends, Growth Factors, & Opportunities
- 4.1.6. Dry Bacterial Nanocellulose (Freeze-Dried, Spray-Dried, Aerogel)
- 4.1.7. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 4.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.9. Key Market Trends, Growth Factors, & Opportunities
- 4.1.10. Functionalized Bacterial Nanocellulose (Surface-Modified, Composite)
- 4.1.11. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 4.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.13. Key Market Trends, Growth Factors, & Opportunities
- 4.1.14. Other Forms (Nanocrystalline, Nanofibrillated Suspension)
- 4.1.15. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 4.1.16. Comparative Revenue Analysis, By Country, 2025 & 2035
- 4.1.17. Key Market Trends, Growth Factors, & Opportunities
- 4.1. Form Market Overview, By Form Segment
- Chapter 5. Bacterial Nanocellulose Market – By Source
- 5.1. Source Market Overview, By Source Segment
- 5.1.1. Bacterial Nanocellulose Market Revenue Share, By Source, 2025 & 2035
- 5.1.2. Komagataeibacter xylinus (formerly Acetobacter xylinum)
- 5.1.3. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 5.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.5. Key Market Trends, Growth Factors, & Opportunities
- 5.1.6. Agrobacterium tumefaciens
- 5.1.7. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 5.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.9. Key Market Trends, Growth Factors, & Opportunities
- 5.1.10. Sarcina ventriculi
- 5.1.11. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 5.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.13. Key Market Trends, Growth Factors, & Opportunities
- 5.1.14. Other Sources (Rhizobium leguminosarum, Pseudomonas fluorescens, Engineered Strains)
- 5.1.15. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 5.1.16. Comparative Revenue Analysis, By Country, 2025 & 2035
- 5.1.17. Key Market Trends, Growth Factors, & Opportunities
- 5.1. Source Market Overview, By Source Segment
- Chapter 6. Bacterial Nanocellulose Market – By Application
- 6.1. Application Market Overview, By Application Segment
- 6.1.1. Bacterial Nanocellulose Market Revenue Share, By Application, 2025 & 2035
- 6.1.2. Biomedical & Healthcare (Wound Dressings, Tissue Engineering, Drug Delivery, Diagnostics)
- 6.1.3. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 6.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.5. Key Market Trends, Growth Factors, & Opportunities
- 6.1.6. Food & Beverage (Texturizer, Stabilizer, Fat Replacer, Dietary Fiber)
- 6.1.7. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 6.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.9. Key Market Trends, Growth Factors, & Opportunities
- 6.1.10. Cosmetics & Personal Care (Skin Hydration, Mask, Active Delivery)
- 6.1.11. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 6.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.13. Key Market Trends, Growth Factors, & Opportunities
- 6.1.14. Electronics & Sensors (Flexible Electronics, Biosensors, Energy Storage)
- 6.1.15. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 6.1.16. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.17. Key Market Trends, Growth Factors, & Opportunities
- 6.1.18. Environmental Remediation (Heavy Metal Removal, Water Purification)
- 6.1.19. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 6.1.20. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.21. Key Market Trends, Growth Factors, & Opportunities
- 6.1.22. Paper & Packaging (Barrier Coatings, Composite Reinforcement)
- 6.1.23. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 6.1.24. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.25. Key Market Trends, Growth Factors, & Opportunities
- 6.1.26. Other Applications (Acoustic Materials, Filtration)
- 6.1.27. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 6.1.28. Comparative Revenue Analysis, By Country, 2025 & 2035
- 6.1.29. Key Market Trends, Growth Factors, & Opportunities
- 6.1. Application Market Overview, By Application Segment
- Chapter 7. Bacterial Nanocellulose Market – By End-Use Industry
- 7.1. End-Use Industry Market Overview, By End-Use Industry Segment
- 7.1.1. Bacterial Nanocellulose Market Revenue Share, By End-Use Industry, 2025 & 2035
- 7.1.2. Healthcare & Pharmaceuticals
- 7.1.3. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 7.1.4. Comparative Revenue Analysis, By Country, 2025 & 2035
- 7.1.5. Key Market Trends, Growth Factors, & Opportunities
- 7.1.6. Food & Beverage
- 7.1.7. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 7.1.8. Comparative Revenue Analysis, By Country, 2025 & 2035
- 7.1.9. Key Market Trends, Growth Factors, & Opportunities
- 7.1.10. Consumer Goods (Cosmetics, Personal Care)
- 7.1.11. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 7.1.12. Comparative Revenue Analysis, By Country, 2025 & 2035
- 7.1.13. Key Market Trends, Growth Factors, & Opportunities
- 7.1.14. Electronics
- 7.1.15. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 7.1.16. Comparative Revenue Analysis, By Country, 2025 & 2035
- 7.1.17. Key Market Trends, Growth Factors, & Opportunities
- 7.1.18. Environmental & Industrial
- 7.1.19. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 7.1.20. Comparative Revenue Analysis, By Country, 2025 & 2035
- 7.1.21. Key Market Trends, Growth Factors, & Opportunities
- 7.1.22. Other Industries (Research, Defense, Textiles)
- 7.1.23. Bacterial Nanocellulose Share Forecast, By Region (USD Billion)
- 7.1.24. Comparative Revenue Analysis, By Country, 2025 & 2035
- 7.1.25. Key Market Trends, Growth Factors, & Opportunities
- 7.1. End-Use Industry Market Overview, By End-Use Industry Segment
- Chapter 8. Bacterial Nanocellulose Market – Regional Analysis
- 8.1. Bacterial Nanocellulose Market Overview, By Region Segment
- 8.1.1. Global Bacterial Nanocellulose Market Revenue Share, By Region, 2025 & 2035
- 8.1.2. Global Bacterial Nanocellulose Market Revenue, By Region, 2026 – 2035 (USD Billion)
- 8.1.3. Global Bacterial Nanocellulose Market Revenue, By Form, 2026 – 2035
- 8.1.4. Global Bacterial Nanocellulose Market Revenue, By Source, 2026 – 2035
- 8.1.5. Global Bacterial Nanocellulose Market Revenue, By Application, 2026 – 2035
- 8.1.6. Global Bacterial Nanocellulose Market Revenue, By End-Use Industry, 2026 – 2035
- 8.2. North America
- 8.2.1. North America Bacterial Nanocellulose Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 8.2.2. North America Bacterial Nanocellulose Market Revenue, By Form, 2026 – 2035
- 8.2.3. North America Bacterial Nanocellulose Market Revenue, By Source, 2026 – 2035
- 8.2.4. North America Bacterial Nanocellulose Market Revenue, By Application, 2026 – 2035
- 8.2.5. North America Bacterial Nanocellulose Market Revenue, By End-Use Industry, 2026 – 2035
- 8.2.6. U.S. Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.2.7. Canada Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.2.8. Mexico Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.2.9. Rest of North America Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.3. Europe
- 8.3.1. Europe Bacterial Nanocellulose Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 8.3.2. Europe Bacterial Nanocellulose Market Revenue, By Form, 2026 – 2035
- 8.3.3. Europe Bacterial Nanocellulose Market Revenue, By Source, 2026 – 2035
- 8.3.4. Europe Bacterial Nanocellulose Market Revenue, By Application, 2026 – 2035
- 8.3.5. Europe Bacterial Nanocellulose Market Revenue, By End-Use Industry, 2026 – 2035
- 8.3.6. Germany Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.7. France Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.8. U.K. Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.9. Russia Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.10. Italy Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.11. Spain Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.12. Netherlands Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.3.13. Rest of Europe Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.4. Asia Pacific
- 8.4.1. Asia Pacific Bacterial Nanocellulose Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 8.4.2. Asia Pacific Bacterial Nanocellulose Market Revenue, By Form, 2026 – 2035
- 8.4.3. Asia Pacific Bacterial Nanocellulose Market Revenue, By Source, 2026 – 2035
- 8.4.4. Asia Pacific Bacterial Nanocellulose Market Revenue, By Application, 2026 – 2035
- 8.4.5. Asia Pacific Bacterial Nanocellulose Market Revenue, By End-Use Industry, 2026 – 2035
- 8.4.6. China Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.7. Japan Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.8. India Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.9. New Zealand Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.10. Australia Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.11. South Korea Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.12. Taiwan Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.4.13. Rest of Asia Pacific Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.5. The Middle-East and Africa
- 8.5.1. The Middle-East and Africa Bacterial Nanocellulose Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 8.5.2. The Middle-East and Africa Bacterial Nanocellulose Market Revenue, By Form, 2026 – 2035
- 8.5.3. The Middle-East and Africa Bacterial Nanocellulose Market Revenue, By Source, 2026 – 2035
- 8.5.4. The Middle-East and Africa Bacterial Nanocellulose Market Revenue, By Application, 2026 – 2035
- 8.5.5. The Middle-East and Africa Bacterial Nanocellulose Market Revenue, By End-Use Industry, 2026 – 2035
- 8.5.6. Saudi Arabia Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.5.7. UAE Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.5.8. Egypt Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.5.9. Kuwait Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.5.10. South Africa Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.5.11. Rest of the Middle East & Africa Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.6. Latin America
- 8.6.1. Latin America Bacterial Nanocellulose Market Revenue, By Country, 2026 – 2035 (USD Billion)
- 8.6.2. Latin America Bacterial Nanocellulose Market Revenue, By Form, 2026 – 2035
- 8.6.3. Latin America Bacterial Nanocellulose Market Revenue, By Source, 2026 – 2035
- 8.6.4. Latin America Bacterial Nanocellulose Market Revenue, By Application, 2026 – 2035
- 8.6.5. Latin America Bacterial Nanocellulose Market Revenue, By End-Use Industry, 2026 – 2035
- 8.6.6. Brazil Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.6.7. Argentina Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.6.8. Rest of Latin America Bacterial Nanocellulose Market Revenue, 2026 – 2035 (USD Billion)
- 8.1. Bacterial Nanocellulose Market Overview, By Region Segment
- Chapter 9. Competitive Landscape
- 9.1. Company Market Share Analysis – 2025
- 9.1.1. Global Bacterial Nanocellulose Market: Company Market Share, 2025
- 9.2. Global Bacterial Nanocellulose Market Company Market Share, 2024
- 9.1. Company Market Share Analysis – 2025
- Chapter 10. Company Profiles
- 10.1. Axcelon Biopolymers Corporation
- 10.1.1. Company Overview
- 10.1.2. Key Executives
- 10.1.3. Product Portfolio
- 10.1.4. Financial Overview
- 10.1.5. Operating Business Segments
- 10.1.6. Business Performance
- 10.1.7. Recent Developments
- 10.2. Hainan Yida Food Industrial Group Co. Ltd.
- 10.3. Cellutech AB
- 10.4. Nanovia SAS
- 10.5. BioMedical Systems Inc.
- 10.6. Nanotech Finland Oy
- 10.7. Bowil Biotech
- 10.8. Nanologica AB
- 10.9. Creative Medical Technology Holdings Inc.
- 10.10. BNC Lab (South Korea)
- 10.11. Biofabrication Lab (Brazil)
- 10.12. Others.
- 10.1. Axcelon Biopolymers Corporation
- Chapter 11. Research Methodology
- 11.1. Research Methodology
- 11.2. Secondary Research
- 11.3. Primary Research
- 11.3.1. Analyst Tools and Models
- 11.4. Research Limitations
- 11.5. Assumptions
- 11.6. Insights From Primary Respondents
- 11.7. Why Custom Market Insights
- Chapter 12. Standard Report Commercials & Add-Ons
- 12.1. Customization Options
- 12.2. Subscription Module For Market Research Reports
- 12.3. Client Testimonials
List Of Figures
Figures No 1 to 39
List Of Tables
Tables No 1 to 51
Prominent Player
- Axcelon Biopolymers Corporation
- Hainan Yida Food Industrial Group Co. Ltd.
- Cellutech AB
- Nanovia SAS
- BioMedical Systems Inc.
- Nanotech Finland Oy
- Bowil Biotech
- Nanologica AB
- Creative Medical Technology Holdings Inc.
- BNC Lab (South Korea)
- Biofabrication Lab (Brazil)
- Others
FAQs
The key players in the market are Axcelon Biopolymers Corporation, Hainan Yida Food Industrial Group Co. Ltd., Cellutech AB, Nanovia SAS, BioMedical Systems Inc., Nanotech Finland Oy, Bowil Biotech, Nanologica AB, Creative Medical Technology Holdings Inc., BNC Lab (South Korea), Biofabrication Lab (Brazil), Others.
Government laws have an effect on Bacterial nanocellulose in the market on a wide variety of levels – between regulatory medical device approval systems that dictate commercial wound care and tissue engineering product access, to regulatory food ingredient approval systems that dictate the quality documentation needed to commercially use BNC, to regulatory pharmaceutical excipient decision systems that dictate the quality documentation needed to commercially adopt BNC. The U.S. regulatory pathway by which commercial BNC wound care and tissue engineering products must pass is the FDA medical device regulatory framework, whereby, under the predicate device pathway, BNC wound dressings have been precleared (via 510(k) premarket clearance) as Class II medical devices, or under the de novo regulatory framework of the new BNC biomedical application under which XCellon currently is registered, where the active submission of XCellon to 510(k) premarket clearance is the most commercially qualified near-term regulatory action. The EU Medical Device Regulation (MDR 2017/745) – that has greatly increased the clinical evidence and performance evaluation requirements on medical devices sold within Europe – places greater regulatory demands on BNC wound dressing and tissue engineering scaffolds seeking CE marking as compared to the prior Medical Device Directive, and it is in this regard that BNC developers are pursuing the generation of substantial clinical data packs in support of CE mark applications that could meet the improved clinical evidence standards of MDR. The FDA GRAS (Generally Recognized as Safe) notification program, through which manufacturers of food ingredients post scientific evidence to support a determination that a food ingredient is generally recognized as safe in its intended use, is the main U.S. regulatory route to commercialization of BNC food ingredients with GRAS notification submissions of BNC in specified food uses currently underway preparations with a number of BNC manufacturers who will provide the regulatory basis upon which BNC food ingredient commercial use can be deployed in the United States. BNC food application regulatory status in Europe is governed by the European Novel Food Regulation (EU) 2015/2283 (which enforces pre-market safety approval of novel food ingredients such as a new source of food additives) and the scientific opinion procedure of the European Food Safety Authority, which is the safety assessment facilitating the affordability of novel food ingredient authorization and demands complete toxicological characterization of food-grade BNC, which responsible BNC producers are increasingly producing. The institutional structure under which the requirements of the bacterial cellulose pharmaceutical-grade quality requirements of pharmaceutical excipient quality standards will be formalized is the monograph development program by the European Pharmacopoeia Commission – the institutional program that sets formal quality standards of pharmaceutical excipients to be used in drug products in Europe – the stated objective of the BioNanoCel Horizon Europe consortium of producing a bacterial cellulose Pharmacopoeia monograph would represent a direct interaction with this regulatory standardization approach, the successful completion of which would have a ground.
The pricing of bacterial nanocellulose has a remarkable range of pricing that covers the application, form, grade of purity, and scale of production. Research-grade BNC (laboratory grade) – small-scale production of BNC in research applications costs USD 500-USD 5000 per kilogram of dry equivalent because of the expensive cost of labor and material of small-scale fermentation and purification of BNC that is incurred in academic and early commercial BNC programs. Food-grade wet BNC on a commercial scale, optimized to achieve large-scale fermentation of low-cost carbon sources, such as commercially available wet food-grade BNC, produced by Hainan Yida and other producers of similar food ingredients, is priced at USD 10.50-USD 50 per kilogram wet weight (equivalent to USD 100-500 per kilogram dry weight). This is a product of this price range in which BNC may compete as a premium functional food ingredient in formulas where the functionality warrants the premium over the Cosmetic grade BNC sourced at USD 200-USD 2,000/kg dry equivalent and grade. BNC is available in premium nanofiber suspension concentrates in high-performance cosmetic applications, which are priced at the top end of the range. At present commercial production scales, medical-grade BNC wound dressings that are made under the ISO 13485 system of quality management systems under sterile processing and endotoxin validation are currently priced between USD 15–USD 80 per unit piece of 10×10 cm wound dressing in the advanced wound care market segment where health economic analysis of superior wound recovery can justify chronic wound care protocols. BNC-based drug delivery, excipient Pharmaceutical grade, about USD 5,000-USD 50,000 per kg in development quantities; Pharmaceutical grade, about USD 5,000-USD 50,000 per kg in commercial production quantities Pharmaceutical-grade BNC-based drug delivery and excipient applications need the manufacturing production to be GMP, pharmacopoeia-compliant, and needs to be characterised Pharmaceutical-grade Pharmaceutical-grade BNC- The main market development facilitator of pricing enhancement is that the cost of medical- and food-grade BNC can be reduced by fermentation scale-up and process cost optimization – with costs of production decreasing by 50-70% at commercial scale growth – allowing an increased variety of applications where BNC cost-performance economics is favorable to conventional materials.
According to present analysis, the market is expected to grow to an approximate USD 1.48 billion by 2035, as the commercialization of BNC wound care products scales over the years and the-out-of-niche specialty to mainstream advanced wound care adoption, as FDA and CE-mark regulatory approval progress, clinical evidence base expands, and cost of manufacture of BNC drops and enables pricing to be competitive with traditional high-end wound dressings, with BNC application in cosmetic manufacturing scaling to premium skincare world wide as Cellutech European distribution, Korean cosmetic brands’ Asian deployment, and Japanese cosmetic company adoption collectively create growing consumer awareness and demand pull for BNC-containing premium skincare products, BNC flexible electronics applications transitioning from research to early commercial deployment as flexible sensor and biodegradable electronics products incorporating BNC reach initial commercial launch at electronics companies and specialized startups, and the progressive development of pharmaceutical-grade BNC as a controlled release excipient expanding BNC into the highest-value pharmaceutical application segment as Pharmacopoeia monograph development and regulatory master file submissions create the documentation infrastructure enabling pharmaceutical formulation adoption, at a CAGR of 14.9% from 2026 to 2035.
North America is expected to maintain the highest revenue share throughout the forecast period, commanding approximately 36% of global market share in 2025, based on the United States’ position as the world’s largest advanced wound care market, representing the highest-value commercial opportunity for BNC wound dressings, the FDA’s established 510(k) regulatory pathway providing commercial approval confidence for BNC medical device developers, the concentration of global pharmaceutical R&D investment in the United States, creating the largest addressable market for pharmaceutical-grade BNC excipient and drug delivery applications, the U.S. biomaterials research ecosystem at MIT, Harvard, Johns Hopkins, and the national laboratory network driving BNC application innovation that creates commercial opportunities across biomedical, electronics, and functional material domains, and Canada’s Axcelon Biopolymers representing the most commercially advanced dedicated BNC medical device developer globally, whose commercial scale-up will establish North America as the leading BNC wound care production center.
Asia Pacific has the highest projected CAGR of 17.8% within the forecast period because China is estimated to have a USD 52 billion BNC market with a CAGR of 19.4% in the commercial production of BNC wound dressing items as Hainan Yida commercializes its products and acquires commercial scale in the manufacturing of wound dressing products with BNC and because China and Japan are rapidly growing advanced wound care markets that will demand commercial BNC wound dressing products and because the electronics manufacturing base in Asia will provide a commercial proximity factor in the development and commercial scale-up of BNC flexible electronics application development and manufacturing scale-up.
The Global Bacterial Nanocellulose Market is estimated to reach massive growth rates in the next few years owing to published epidemiology estimating 2.5% of the U.S. population having a chronic wound at any specific time as representative of about 8.2 million Americans with a large addressable wound care market in which BNC clinical performance benefits can be converted to commercial opportunity, the commercial scale production of BNC food ingredients that has already demonstrated manufacturing feasibility of low-cost large-scale production of BNC, the 3D printing of bacteria-containing BNC composite product development, Axcelon’s XCell® clinical data demonstrating 68% complete healing at 12 weeks in venous leg ulcer patients versus 42% for conventional treatment providing quantified clinical superiority evidence supporting healthcare adoption, Hainan Yida’s commercial-scale BNC food ingredient production demonstrating the manufacturing feasibility of cost-effective large-scale BNC production that is enabling food and cosmetic application economics, Cellutech’s CelluForce demonstrating 340% improvement in skin hydration retention over 8 hours in clinical testing providing quantified cosmetic performance evidence supporting premium skincare adoption, the flexible electronics market’s demand for biodegradable substrate materials that BNC uniquely satisfies among natural materials providing structural long-term growth in the fastest-growing application segment, and Empa’s 3D-printable bacteria-containing BNC composite demonstrations providing the research foundation for next-generation customized biomedical applications that will expand the commercial BNC market further beyond current wound care and cosmetic applications.